the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Modeling gas exchange and biomass production in West African Sahelian and Sudanian ecological zones
Jaber Rahimi
Expedit Evariste Ago
Augustine Ayantunde
Sina Berger
Jan Bogaert
Klaus Butterbach-Bahl
Bernard Cappelaere
Jean-Martial Cohard
Jérôme Demarty
Abdoul Aziz Diouf
Ulrike Falk
Edwin Haas
Pierre Hiernaux
David Kraus
Olivier Roupsard
Clemens Scheer
Amit Kumar Srivastava
Torbern Tagesson
West African Sahelian and Sudanian ecosystems provide essential services to people and also play a significant role within the global carbon cycle. However, climate and land use are dynamically changing, and uncertainty remains with respect to how these changes will affect the potential of these regions to provide food and fodder resources or how they will affect the biosphere–atmosphere exchange of CO2. In this study, we investigate the capacity of a process-based biogeochemical model, LandscapeDNDC, to simulate net ecosystem exchange (NEE) and aboveground biomass of typical managed and natural Sahelian and Sudanian savanna ecosystems. In order to improve the simulation of phenology, we introduced soil-water availability as a common driver of foliage development and productivity for all of these systems. The new approach was tested by using a sample of sites (calibration sites) that provided NEE from flux tower observations as well as leaf area index data from satellite images (MODIS, MODerate resolution Imaging Spectroradiometer). For assessing the simulation accuracy, we applied the calibrated model to 42 additional sites (validation sites) across West Africa for which measured aboveground biomass data were available. The model showed good performance regarding biomass of crops, grass, or trees, yielding correlation coefficients of 0.82, 0.94, and 0.77 and root-mean-square errors of 0.15, 0.22, and 0.12 kg m−2, respectively. The simulations indicate aboveground carbon stocks of up to 0.17, 0.33, and 0.54 kg C ha−1 m−2 for agricultural, savanna grasslands, and savanna mixed tree–grassland sites, respectively. Carbon stocks and exchange rates were particularly correlated with the abundance of trees, and grass biomass and crop yields were higher under more humid climatic conditions. Our study shows the capability of LandscapeDNDC to accurately simulate carbon balances in natural and agricultural ecosystems in semiarid West Africa under a wide range of conditions; thus, the model could be used to assess the impact of land-use and climate change on the regional biomass productivity.
- Article
(6633 KB) - Full-text XML
- BibTeX
- EndNote
Land-cover and land-use changes significantly affect water, carbon (C), and energy exchange processes between the biosphere and the atmosphere and, thus, climate change (Massad et al., 2019; Pielke et al., 2011). Within the larger biomes, savanna or semiarid grassland systems have been highlighted as being of particular importance: on the one hand, they are assumed to store large amounts of C (Elberling et al., 2003; Scholes and Hall, 1996); on the other hand, they experience large C exchanges (Ahlström et al., 2015). Additionally, savannas are vulnerable to climate change, specifically to changing rainfall patterns or increasing fire intensity or frequency (Grossiord et al., 2017; Livesley et al., 2011). In particular, the role of West African savanna systems in global C cycling has attracted increasing attention over the last decade (Quenum et al., 2019; Bocksberger et al., 2016; Sjöström et al., 2011) due to considerable changes in climate but also owing to considerable changes in land cover, such as the extension of agriculture and the intensification of forest logging (Odekunle et al., 2008). These changes may already have and will further affect the C exchange rates between semiarid West African savanna ecosystems and the atmosphere, which might not only affect biomass production but may also threaten biodiversity as well as the livelihood of people (Dimobe et al., 2018; Hartley et al., 2016; Dayamba et al., 2016).
Hence, in order to better understand the impacts of climate and land-use change on biosphere–atmosphere interactions across West Africa, it is important to improve our understanding of ecosystem C cycling and C exchange with the atmosphere. This can be done by using existing knowledge to set up ecosystem models and to test if these models are able to (a) realistically represent the sensitive responses of semiarid ecosystems to climatic variation and land-use management, and (b) accurately represent C exchange processes as well as (c) the distribution of above- and belowground C pools.
Several modeling approaches, which concentrated on land-use management (Boone et al., 2004) or population dynamics (Tews et al., 2006; Tews and Jeltsch, 2004), have been used to describe savanna ecosystem processes and semiarid cropland development over the past decades. If physiological processes were covered, they were treated with very coarse resolution (Scheiter and Higgins, 2009) or for herbaceous species only (Delon et al., 2019; Grote et al., 2009b). Few terrestrial modeling approaches have been evaluated against field observations in order to assess their ability to simulate ecosystem C dynamics and the biosphere–atmosphere exchange of savanna-type ecosystems (e.g., Whitley et al., 2016, 2017). It has been specifically highlighted that it is crucial to represent the plant phenological development for semiarid systems with strong seasonal variations in plant water availability, which is something that most models fail to achieve (Pitman, 2003). Indeed, the phenology of deciduous plants including most crops is usually related to temperature development, which is generally the limiting factor in temperate ecosystems (Vitasse et al., 2011; Wesolowski and Rowinski, 2006). For tropical (and sometimes also boreal) regions, which are assumed to be evergreen, phenology is often forced or completely neglected (Mougin et al., 1995; Saleska et al., 2003). In savannas, however, the growth of new tissues in grass and trees is often related to the onset of the rainy season, indicating that water availability is a crucial determinant (Kucharik et al., 2006). Models that account for this influence are still rare. However, water supply has been included in a few models as a direct impact on budburst, which defines the start of growth for grasses in semiarid regions (Akponikpè et al., 2010; Ivanov et al., 2008; Loustau et al., 1992). Moreover, Yuan et al. (2007) used simulated soil moisture values as a driver to modify the phenological development of the grass layer of a semiarid steppe, and Jolly and Running (2004) defined the onset of budburst for a grass-dominated Kalahari ecosystem as the date on which daily precipitation exceeded potential evaporation and the start of leaf senescence as the point at which soil moisture falls below a defined threshold. Nevertheless, while important for grasses, most trees do not seem to be bound to a minimum water content of the upper soil layers for leaf flushing, possibly because they have access to deeper water reserves (Do et al., 2005).
Thus, the simulation of C balances in semiarid tropical systems such as those found in West Africa requires a close and consistent link between rainfall, soil, and vegetation processes (Boke-Olén et al., 2016). Moreover, as many of the ecosystem types found in this region are composed of a tree–shrub layer and a layer of ground vegetation (mainly grasses, sometimes also crops), models should be able to distinguish between different vegetation types and their competition for light and other resources. In addition, it needs to be considered that many of the savanna ecosystems are used by pastoralists or mixed crop–livestock farmers for grazing and cropping (Ker, 1995). Thus, these management options should also be represented in the model.
In this study, we parameterized, complemented, and evaluated LandscapeDNDC (Haas et al., 2013), a model framework that uses the soil C processes, nitrogen (N) processes, and water processes derived from the original DNDC (DeNitrification-DeComposition) model (Li et al., 1992). The framework can be combined in a flexible manner with cohort-based ecosystem models (Grote et al., 2011b) as well as crop growth models (Kraus et al., 2016). So far, the model framework and its predecessors have been used and applied to various natural and managed temperate and tropical ecosystems such as forests, grasslands, or rice paddies (Kraus et al., 2016), including savanna grasslands (Grote et al., 2009b). However, to date, it has never been tested for its suitability to simulate C fluxes over a wide range of different managed and natural savanna ecosystems including different vegetation types.
For this analysis, a literature review was first carried out to find suitable parameters for the physiological processes of typical grass and tree species in Sahelian and Sudanian savanna ecosystems. Furthermore, thresholds of soil-water availability for leaf development and senescence, which were derived from 12 sites with ample data available for several years, were introduced. Finally, the model was evaluated against data from other sites representative of the region, including eddy flux measurements, satellite data, and in situ biomass measurements for both managed and natural ecosystems.
The objective of this paper is to test the ability of the LandscapeDNDC model in its current form to simulate C fluxes and stocks for various representative savanna ecosystem types with varying human management activities within the Sahelian and Sudanian regions.
2.1 Study area
West African semiarid drylands are located between 15∘ E–16∘ W and 7–19∘ N (between the Sahara and the Guinean zone) and are spread over 11 countries (Benin, Burkina Faso, Côte d'Ivoire, Gambia, Ghana, Guinea, Mali, Niger, Nigeria, Senegal, and Togo; Fig. 1). Following a transect of decreasing precipitation from south to north, there is a gradual transition from forest, woodland, savanna woodland, savanna grassland, to semi-desert grassland (Kaptue Tchuente et al., 2010). Along this gradient, the degree of ground cover and the proportion of woody species (trees, shrubs, and bushes) decreases and the vegetation becomes shorter. This region is classified into two distinct ecological zones, “Sahelian” and “Sudanian”, which differ mainly in terms of precipitation amount and dry season length. In the Sahelian ecological zone which extends over 1.3×106 km2, the average annual temperatures vary between 25 and 31 ∘C and the annual precipitation is between 150 and 600 mm. The length of dry season lasts for 7–9 months annually and the monthly precipitation maximum is in August. The Sudanian zone, which covers approximately 1.7×106 km2, is cooler (22–29 ∘C), wetter (600–1200 mm yr−1), and the dry season length is around 4–7 months with monthly maximum precipitation also occurring around August (NASA POWER climate dataset, https://power.larc.nasa.gov/, last access: 16 June 2021).
2.2 Datasets for simulation and evaluation
2.2.1 Initialization, climate, and deposition
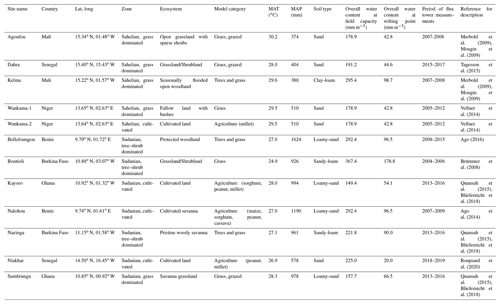
As model input data, initial soil and vegetation properties as well as daily climate data are required. Land management needs to be prescribed as boundary conditions. The soil parameters were bulk density (kg m−3), pH, soil texture (i.e., clay, silt, and sand content), organic C and N content (kg kg−1), and soil hydrological parameters (i.e., field capacity, wilting point; in mm m−3). These were gathered from available literature sources for each site and complemented by information from the principal investigators (PI) of these sites. In a few cases, soil information was complemented by data from ISRIC-WISE (International Soil Reference and Information Centre – World Inventory of Soil Emission Potentials) soil dataset (Batjes, 2008). Similarly, vegetation was initialized using data of the amount of grass biomass, tree sizes (average height and breast height diameter), and number of trees per hectare as recorded at the sites.
Input climate data were maximum and minimum temperature (∘C), precipitation (mm), relative humidity (%), solar radiation (W m−2), and wind speed (m s−1). These data were either obtained directly from measurements at the sites or were sourced from the NASA POWER climate dataset. Except for Kelma, all sites were assumed to have no ground water access down to their maximum rooting depth of 1 m. For the Kelma site, a flooding period was prescribed for 2005–2007, according to published water content data (Timouk et al., 2009; de Rosnay et al., 2009). Management was also prescribed for both agricultural sites and savanna grasslands that were occasionally grazed with cattle. Fire impacts were not considered.
In addition to climate, daily input of dry and wet deposition of oxidized and reduced N compounds (nitrate, ammonia, nitric acid, and nitrogen oxide) were provided for the model runs. These were derived from the field measurements within the IGAC/DEBITS/AFRICA (International Global Atmospheric Chemistry/Deposition of Biogeochemically Important Trace Species/AFRICA) project, in which some of our investigation sites were also included (Agoufou, Banizoumbou, Djougou, and Katibougou). According to this dataset, the total wet deposition of N was estimated to be around 3.2 kg, and dry deposition was estimated to be around 3.6 kg N ha−1 yr−1, with only little variation between Sahelian and Sudanian ecosystems (Galy-Lacaux et al., 2014).
2.2.2 Carbon flux and soil moisture measurements
We collected net ecosystem C exchange (NEE) measurements from 12 flux tower sites, hereinafter called the core sites, in the Sudanian and Sahelian ecological zones. These data were used for calibrating the new phenological routine of the LandscapeDNDC model and to evaluate simulations for crop-dominated, tree-dominated grassland/woodland, and grass-dominated ecosystems. These eddy-covariance measurements were carried out within the CARBOAFRICA (http://www.carboafrica.eu, last access: 16 June 2021), AMMA-CATCH (African Monsoon Multidisciplinary Analysis – Coupling the Tropical Atmosphere and the Hydrological Cycle) observatory (http://www.amma-catch.org, last access: 16 June 2021), and AMMA (http://www.amma-international.org, last access: 16 June 2021) projects, or by the WASCAL (West African Science Service Centre on Climate Change and Adapted Land Use) research center (http://wascal.org, last access: 16 June 2021).
The spatial distribution and characteristic of the core sites show that they are well distributed across the major natural and agricultural areas in the Sudanian and Sahelian ecological zones covering grassland savannas (from sparse grasslands to shrublands), woodlands (including seasonally flooded, open, and dense forests), and cultivated land types (with major crops including maize, Zea mays; millet, Pennisetum glaucum; sorghum, Sorghum bicolor; peanut, Arachis hypogaea; and cassava, Manihot esculenta (Fig. 1; Table 1). Five sites are located in the Sahelian ecological zone (Dahra, Senegal; Kelma, Mali; Agoufou, Mali; Wankama-1 and -2, Niger), and seven in the Sudanian ecological zone (Sumbrungu, Ghana; Bontioli, Burkina Faso; Kayoro, Ghana; Nalohou, Benin; Nazinga, Burkina Faso; Niakhar, Senegal; Bellefoungou, Benin). For more information, see Table 1 and the references therein.
In addition, soil moisture data measured close to the flux towers were used for evaluation of simulations. These measurements took place mostly within the first 30 cm (CS616 soil moisture probes, Campbell Scientific Inc., USA), although deeper soil horizons were also considered at the AMMA sites. In few cases, only moisture of the uppermost layers were recorded for comparison with estimates from satellite observations. Thus, in order to be consistent across sites, we use values recorded for the first 10 cm, which are also those with the most complete records.
2.2.3 Aboveground biomass and leaf area measurements
Aboveground herbaceous biomass and crop yield were available from sampled measurements for all core sites except for two of the Sudanian cultivated sites (Kayoro and Nalohou). For the Kayoro site, biomass was assumed to equal that at the Vea site in Ghana, and for the Nalohou site, maize data were taken from the Dassari site in Benin and the Wa site in Ghana, sorghum was taken from the Samanko site in Mali, and cassava was taken from the Ikenne site in Niger, because these sites have a similar climate.
In addition, 42 sites representing cropland (11), grassland savanna (27), and grassland ecosystems with significant tree contributions (4) from which biomass data are available were used for validating the revised model (Fig. 1). These sites are located in Senegal, Benin, Mali, Ghana, Niger, and Burkina Faso covering overall 70 years of observations within the 1984–2018 period (not continuous for all sites). A total of 24 sites in Senegal (20 grassland and 4 savanna woodlands) that were monitored between 1999 and 2017 were provided by the Centre de Suivi Ecologique (CSE; Diouf et al., 2015). Data from four sites with contributions of trees as well as grasses (Eguerit, Hombori Hondo, Tara, and Timbadior) and one millet site (Bilantao) were obtained from the Gourma region in Mali, representing a precipitation gradient from 300 to 450 mm yr−1. These field measurements cover the period between 2005 and 2016 and were gathered within the African Monsoon Multidisciplinary Analysis (Mougin et al., 2019, 2009). Three more fallow and grassland sites are located in the Fakara region in western Niger (Banizoumbou, Tigo Tégui, and Kodey). Measurements here began in 1994, taken by the International Livestock Research Institute (ILRI; Hiernaux and Ayantunde, 2004), and continued until 1999 under the responsibility of the AMMA-CATCH observatory (Cappelaere et al., 2009). Furthermore, we gathered biomass production and yield data covering all major crops of the core sites from three sources. The first dataset, containing data for maize, millet, and sorghum for 2004, 2005, 2010, 2015, and 2016 (with different fertilization treatments and different crop-sowing windows), consists of one site in Benin (Dassari), two sites in Ghana (Vea and Wa), and sites in Mali (Samanko) and Niger (Sadore) (Faye et al., 2018). The second dataset, obtained in Upper Ouémé catchment in the Republic of Benin (for 2001, 2002, and 2003), provides information on total aboveground biomass for three sites (Dogue, Beterou, and Wewe) for peanut, maize, and sorghum production with different managements (e.g., different fertilization types/amounts) (Dagbenonbakin, 2005). The third dataset, used for validating LandscapeDNDC, was from cassava fields from the Ikenne and Oke-Oyi sites in Nigeria (Sobamowo, 2016).
Leaf area index (LAI; the one-sided leaf area per unit ground area) data for comparison with measurements were taken at a 4 d temporal and a 500 m spatial resolution from the MODerate resolution Imaging Spectroradiometer (MODIS) satellite data (MCD15A3H; Myneni et al., 2015). Only pixels where the core sites are located were used. Previous investigations of MODIS data have found them to be the most suitable for representing the seasonality of vegetation dynamics (Gessner et al., 2013; Mougin et al., 2019). We undertook an additional investigation using any data that field site managers could provide, and our findings generally supported the abovementioned view (Fig. 2). However, MODIS data are somewhat higher than site measurements when LAI is small and are lower when LAI is large. The reasons for this include particularly large fractions of missing data during high-LAI episodes (Gessner et al., 2013), the size of the pixels that often include a tree contribution at sites treated as agricultural or grassland sites here (Kahiu and Hanan, 2018), as well as other specific reasons that are addressed in the discussions of the results for the specific sites.
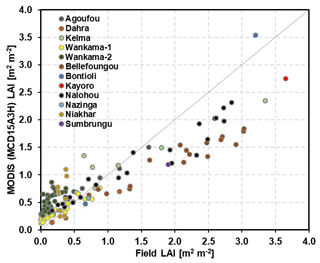
Figure 2Comparison between field LAI measurements and MODIS (MCD15A3H) LAI estimation at different eddy-covariance sites.
2.3 The LandscapeDNDC model
2.3.1 Description and parameterization
LandscapeDNDC is a framework for one-dimensional biogeochemical models, which mainly simulate C, water, and N cycling between the atmosphere, vegetation, and soil at daily to sub-daily temporal resolution for various ecosystems, i.e., arable, grassland, and forest (Haas et al., 2013). We used version 1.30.4 of this model (https://ldndc.imk-ifu.kit.edu/, last access: 16 June 2021) including the MeTrx sub-model for soil biogeochemistry and soil respiration (Kraus et al., 2015), the ECM sub-model for microclimate (Grote et al., 2009a), and the original DNDC routines to describe the water cycle (Li et al., 1992; Kiese et al., 2011). For grasslands and grass/woodlands, the PSIM physiological simulation module (Grote et al., 2011a; Werner et al., 2012) was used, which has been widely applied for forests, including sites where ground vegetation needed to be considered (Lindauer et al., 2014; Dirnböck et al., 2020), as well as for savanna grasslands (Grote et al., 2009b). However, the model does not consider the production of fruits or similar. Therefore, we applied the PlaMox module (Kraus et al., 2016; Liebermann et al., 2020), which has been developed particularly to described crop growth based on the same physiological processes as PSIM, for the agricultural plants.
While parameters for agricultural crops were mostly defined in a species-specific manner in the model already, information about physiological properties of savanna grass and tree species were newly collected from literature. Some properties were assumed to be ubiquitous for savanna trees and grasses, but we also recognized some species-specific differences between the Sudanian and Sahelian ecosystems. Therefore, we selected Acacia (in particular A. tortilis, also known as Vachellia tortilis) as the dominant tree genus and Cenchrus biflorus as the dominant grass species for the Sahelian zone. For the Sudanian ecoregion, we chose Burkea africana as the dominant tree species and Andropogoneae (in particular Andropogon gayanus) as the dominant grass type (Bocksberger et al., 2016; Geerling, 1985; Sotelo Montes et al., 2014). The parameters describing the specific physiological properties of each species are given in Table 2 for natural species and in Table 3 for agricultural species.
Both vegetation models (PSIM and PlaMox) used the Farquhar approach (with the extension for C4 photosynthesis by Collatz) (Farquhar et al., 1980; Collatz et al., 1992), which requires several parameters related to enzyme activities (also see Tables 2 and 3). Respiration was differentiated into growth respiration and maintenance respiration. Growth respiration was estimated as a fixed fraction of net photosynthesis (25 %). Maintenance respiration was calculated using a linear relationship to N content but was modified by temperature and the relative depletion of C reserves as done by Thornley and Cannell (2000). Carbon was allocated into the different plant tissues according to Grote (1998), with leaf expansion determined by growing degree sum (see below) and C supplied from previous-year storages.
Evapotranspiration was driven by potential evapotranspiration, calculated with a modified Thornthwaite approach (Camargo et al., 1999; Pereira and Pruitt, 2004; Thornthwaite, 1948) that depends on temperature and day length and drives transpiration demand except in periods with wet surfaces. Actual transpiration, however, is constrained by soil-water availability down to rooting depth (weighted by root mass density) as well as transpiration requirements for photosynthesis (calculated using a water use efficiency coefficient, WUEC; see Tables 2 and 3). WUEC is considered to increase with decreasing relative available soil-water content (RWC) for grasses and trees, but it is constant for crops. Without soil-water restrictions, photosynthesis and stomatal conductance were iteratively calculated based on the Ball–Berry approach (Ball et al., 1987) using a scaling parameter (GSA, see Table 2) and the species-specific maximum conductance as parameters (minimum conductance is generally set to 10 mmol H2O m−2 s−1). If relative soil-water availability is lower than a threshold value, either the stomatal conductance (PSIM) or the photosynthesis itself (PlaMox) was linearly reduced (Leuning, 1995; Knauer et al., 2015). The threshold is defined as the species-specific parameter H2OREF_GS or H2OREF_A, respectively (Tables 2, 3). Interception was calculated from LAI, which is the product of specific leaf area and foliage biomass, and specific interception capacity following Gash et al. (1995). For soil-water availability, we assumed a rooting soil depth of 1 m for all sites, with no difference between trees and grasses (according to the ISRIC soil database available at https://www.isric.org/explore/wise-databases, last access: 16 June 2021). Fine-root abundance decreased exponentially with depth, with roots of grasses slightly more concentrated in the upper soil layers than tree roots (February and Higgins, 2010).
Leaf flushing was assumed to begin when a threshold cumulative daily temperature sum (growing degree days) had been reached and persisted for a defined period of days. The newly grown foliage was assumed to be fully shed after a period defined by the leaf longevity, and the shedding also needed a defined period of days (Grote, 2007). In order to account for the close relationship between leaf flushing of savanna grasslands and rainfall events, we included a constraint that foliage only started to grow if the RWC was above a threshold value. Similarly, we also included a restriction so that drought-related senescence could only occur after foliage was at least 90 % developed. All phenological parameters were defined as the best fit to all Sahelian and Sudanian core sites for natural as well as agricultural systems, respectively (Tables 4, 5).
2.3.2 Model setup and initialization
All simulations used a 3-year spin-up to account for an uncertain initialization of carbon and nitrogen pools that are adjusting to general environmental conditions during this time. At agricultural sites, identical planting schemes are deployed with tilling always occurring before the planting date, meaning that all straw and grasses that remained on the field were transferred to the soil and incorporated into the soil C and N pools. No additional irrigation nor fertilization was reported at any site and, thus, is also not simulated. Grasslands are initialized with a total biomass of 1000 kg ha−1 at the beginning of the simulations at all sites and adjusted during spin-up years to a value that accounts for the competition on light and water at the sites.
Agricultural sites
For Kayoro, peanut was planted first each year (June–July), followed by pearl millet or sorghum that was harvested in October. At the Niakhar site, pearl millet was sown on 15 May 2018 (sowing density of 0.8 m−2) and harvested on 12 October. In 2019, peanut was planted on 5 July (sowing density of 8.33 m−2) and harvested at end of November. Wankama-2 had been under continuous millet cultivation, which was generally planted after the start of the wet season (around 10 July; sowing density of around 10 000 pockets per hectare) and harvested when precipitation had ceased (mid-October). At Nalohou, the landscape was more heterogeneous and eddy flux measurements were influenced by both mixed crops and fallow bush. All crops at this site were planted/grown during the wet season (between April and September–October). The results were post-processed according to the footprint analysis (which indicates the contribution of each vegetation type to the measured flux for the wet and dry season), percentage of area planted by each crop, and cropping pattern provided by Ago et al. (2014).
Savanna sites
Grasslands were supposed to be fully covered with either the Sudanian or Sahelian grass type, whereas mixed grass/woodlands were simulated by considering grass and tree species as different cohorts within the same simulation run (Grote et al., 2011b). Therefore, competition effects between the plant groups depended on the abundance of trees which was characterized by a ground cover of 80 % in Bellefoungou, 72 % in Nazinga (Sudanian), and 25 % in Kelma (Sahel). This was initialized by first defining the dimension (height and diameter at 1.3 m) of the average tree at the specific site, calculating the ground cover according to allometric relations described in Grote et al. (2020), and carrying out parametrization with data from the literature (Buba, 2013). These calculations do not assume a difference between species allometry. The total number of trees at the site was adjusted in order to reach the measured total ground cover.
Table 2Plant parameters affecting the C and water exchange in the PSIM vegetation model used for simulating biomass growth of natural savanna ecosystem types.
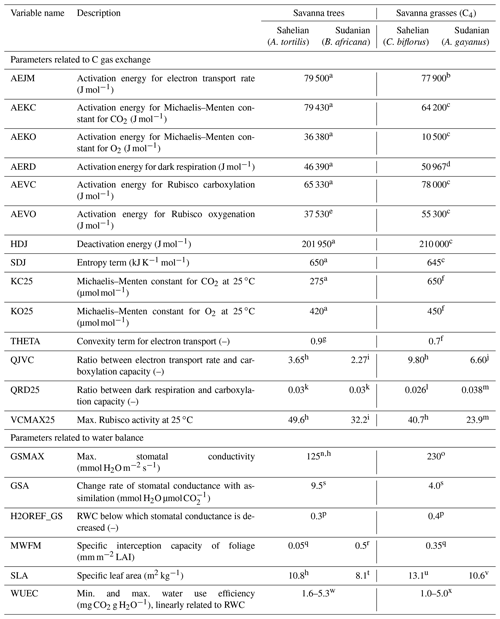
a Chen et al. (2008), b Massad et al. (2007), c Boyd et al. (2015), d Collatz et al. (1992), e Martin et al. (2000), f Von Caemmerer (2000), g Thornley (2002), h Sibret (2018), i Kgope and Musil (2004), j Kim and Verma (1991), k Fürstenau Togashi et al. (2018), l Sonawane et al. (2017), m Feng and Dietze (2013), n Ullmann (1989), o Simioni et al. (2000), p Baldocchi et al. (2004), q de Jong and Jetten (2007), r Running and Coughlan (1988), s Baldocchi and Meyers (1998), t Guenther et al. (1996), u Chandra and Dubey (2008), v Setterfield et al. (2018), w Kebbas et al. (2015), and x Snyman (2005).
Table 3Plant parameters affecting the C and water exchange in the PlaMox vegetation model used for simulating biomass growth and yield at the agricultural sites.
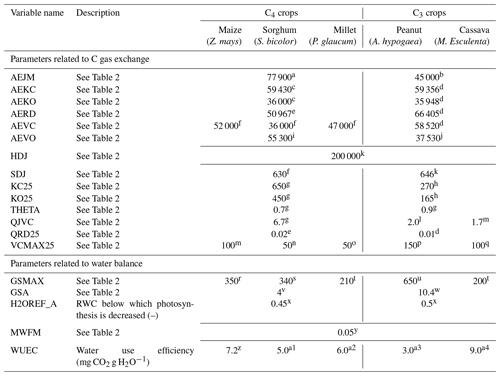
a Massad et al. (2007), b Groenendijk et al. (2011), c Yu et al. (2001), d Farquhar et al. (1980), e Collatz
et al. (1992), f Sonawane et al. (2017), g Von Caemmerer
(2000), h Bernacchi et al. (2002), i Boyd et al. (2015), j Martin et al. (2000),
k Kattge and Knorr (2007), l Ainsworth and Rogers (2007),
m Caldararu et al. (2017), n Vico
and Porporato (2008), o Kothavala et al. (2005), p Pallas and Samish (1974), q de Souza Nóia
Júnior et al. (2020), r Gleason et al. (2017), s Körner et al. (1979), t Da Matta et al. (2001),
u Vu (2005), v Sellers et al. (1996), w Baldocchi and Xu (2005), x from FAO database
(http://www.fao.org/3/X0490E/x0490e0e.htm#total%20available%20water%20(taw), last access: 16 June 2021), y Running and Coughlan (1988), z Holá et
al. (2010), a1 Ünlü and Steduto (2000), a2 Vitkauskaitė and Venskaitytė (2011),
a3 Kalariya et al. (2015), and a4 Yao and Goué
(1992).
As there is a certain variability for this parameter
(H2OREF_A) with respect to species and sites, we conducted an
additional analysis to assess how sensitive biomass production of each crop
species responds to this parameter. To do this, we varied
H2OREF_A from 0.30 to 0.70 at each site and for each
species. Results indicated that when applying the upper limit of 0.7,
productivity was lower than the standard value for C3 (0.5) and C4 (0.45)
crops by 6.4 % for peanut, 3.9 % for millet, 1.9 % for cassava,
0.78 % for sorghum, and 0.11 % for maize. Conversely, applying
the lower limit of 0.3 increased productivity relative to the standard value
by 3.8 % for peanut, 1.5 % for millet, 1.1 % for cassava, 0.07 % for
sorghum, and 0.05 % for maize. Thus, the overall sensitivity of biomass
production to the RWC threshold value of photosynthesis decline was judged
to be low.
Table 4Plant parameters related to new phenological processes used in the PSIM model for describing dynamics in natural savanna ecosystem types, derived from measurements at the core sites of the investigation.

a Mougin et al. (2019), Timouk et al. (2009), and Sjöström et al. (2013); b Quansah et al. (2015) and Ago et al. (2016); c Delon et al. (2015), Hiernaux et al. (2009), Timouk et al. (2009), Baup et al. (2007), Tagesson et al. (2016), Boke-Olén et al. (2016), and Velluet et al. (2014); and d Quansah et al. (2015) and Berger et al. (2019)
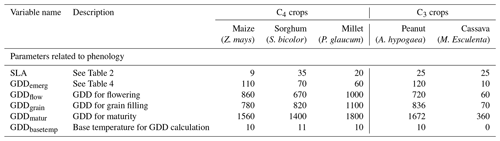
Table 5Plant parameters that describe phenology and ontology/yield in the PlaMox vegetation model for agricultural simulations (taken from the WOFOST model v.6.1 http://github.com/ajwdewit/WOFOST_crop_parameters, last access: 16 June 2021).
2.4 Statistical analysis
To identify the relationship between the simulated and measured NEE and LAI, a Pearson correlation coefficient (r) analysis was performed. The correlation coefficient measures the strength of the linear relationship between two variables (here, between the simulated and measured NEE and LAI during the crop-growing period for the agricultural sites and for the entire year for savanna grasslands and savanna mixed tree–grassland sites).
3.1 Agricultural sites
Figure 3 shows measurements and simulation results for NEE and LAI for the four core sites that are used for agriculture (Kayoro, Niakhar, Nalohou, and Wankama-2). It should be noted that LAI simulations are only shown during the crop-growing period and are otherwise assumed to be zero.
For Kayoro in northern Ghana, the predominant cropping pattern between 2013 and 2016 is peanut in rotation with pearl millet (2013, 2015) or peanut in rotation with sorghum (2014, 2016). Simulated seasonal dynamics and magnitudes of NEE and LAI are well in accordance with measurements (r=0.79 and 0.76, respectively, Fig. 3). However, in 2013, the model overestimated LAI and, therefore, C uptake during the crop-growing period (−55.5 kg C ha−1 d−1 was estimated from measurements, whereas −72.1 kg C ha−1 d−1 was simulated). A possible reason for this may have been the occurrence of weeds, which may have prevented peanut and millet from growing to their full potential, or that the fetch of the eddy-covariance tower extended beyond the investigation area where less productive plants or bare land resulted in a reduction in the average data from measurements (Quansah et al., 2015).
At Niakhar, a representative site for the so-called “groundnut basin” of Senegal, either pearl millet (2018) or peanut (2019) was grown in an annual rotation. LAI for millet and peanut was underestimated by the model, both by approximately 30 %. This is likely due to trees in the region (Roupsard et al., 2020) that are considered in the observations but not simulated. Nevertheless, the deviations between measured and modeled NEE were relatively small (r=0.80). A possible uncertainty at this site is the presence of Faidherbia albida trees (6.8 trees ha−1) that typically show a different phenology which explains a negative NEE (C uptake) during the dry season that was not considered by the simulations.
At Nalohou, located in the Ara watershed in the northern part of Benin, four crops were planted sequentially and periodically and several crops even grew simultaneously, explaining the rather irregular shape of NEE and LAI (including a continuous cover throughout the dry season in 2009). Nevertheless, simulations of both terms generally agree with measurements, showing average correlation coefficients of 0.74 and 0.86, respectively. Uncertainties are related to the vegetation distribution. For example, a footprint analysis presented by Ago et al. (2014) showed that crops contribute only about 77 % of the C exchange fluxes, whereas the rest is attributed to natural savanna. Variation between years also occur due to different climate conditions, such as the precipitation that was about 25 % higher in 2009 than in 2008. The relatively high precipitation, which was above long-term average in both years, may partly explain the insensitivity of NEE to the underestimation of water storage in the simulations. This underestimation mainly originates from a field capacity that has been initialized with a relatively small value based on site measurements and, thus, does not allow for the high moisture values that have actually been observed. It is obvious that the mismatch is caused by the small-scale heterogeneity of upper-soil conditions at this site. Overall, the long vegetation period of Nalohou caused it to become a substantial C sink of 2814 kg C ha−1 yr−1.
For Wankama-2, located in the southwest of the Republic of Niger, annual rotations with pearl millet remained unchanged throughout the 7-year study period (2005–2012). Simulated values were highly correlated with NEE and LAI, with coefficients of 0.84 and 0.82, respectively. Moreover, simulated patterns of biomass development closely matched observations. The simulated C sink was on average 810 kg C ha−1 yr−1, whereas estimates based on measurements were 720 kg C ha−1 yr−1. However, the simulations indicate a larger variability in yields than indicated by measurements, leading, for example, to smaller than average yields during 2009–2010 (0.12 kg m−2 compared with an average of 0.21 kg m−2). These years were somewhat dryer than average (320 mm of annual precipitation in 2009–2010, whereas the normal amount is around 410 mm yr−1), indicating that the model seems to be too sensitive to changes in soil-water availability. Other uncertainties are potentially varying planting densities and the occurrence of weeds.
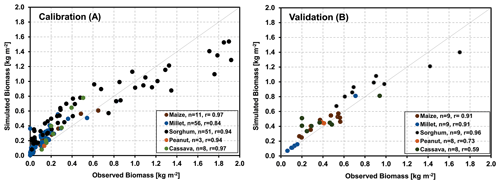
The results of the agricultural core sites were obtained by calibrating the parameters that determine the maximum biomass and yield values separately for all five crops using observations. Accordingly, the deviation between measured and simulated biomass/yield values across all sites has been minimized, resulting in an overall correlation coefficient of 0.93 (Fig. 4a). Simulating biomass production for six further sites (17 years) across West Africa with the derived parameters and no further adjustment yielded an overall correlation coefficient of 0.82 (Fig. 4b), indicating that the model was very suitable for representing the development of the major crops throughout the investigated area.
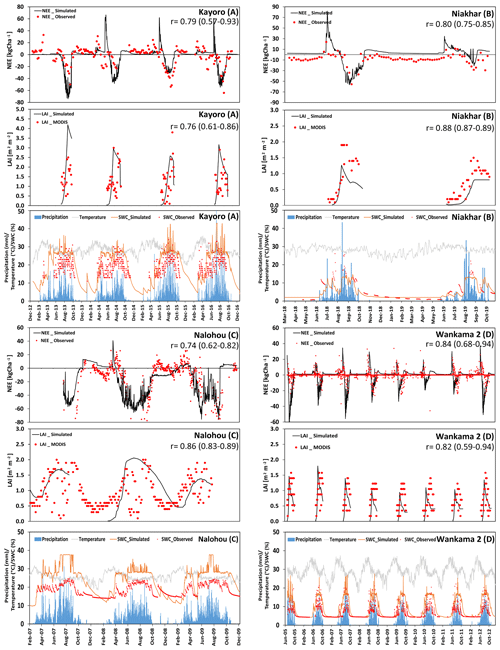
Figure 3(a–d) Time series of the observed and modeled NEE (kg ha−1; negative values represent a flux into the biosphere, and positive values represent a flux towards the atmosphere), LAI (m2 m−2), and soil-water content (at 10 cm depth). In addition, precipitation and temperature at four different crop-dominated sites in the Sahelian (d) and Sudanian (a–c) ecological zones are presented.
3.2 Savanna grasslands
Figure 5 shows the comparison of measured and simulated NEE and LAI for the five grass-dominant core sites used for calibration. They are differentiated according to their location in the Sahelian (panels a–c: Agoufou, Dahra, and Wankama-1) and Sudanian (panels d–e: Sumbrungu and Bontioli) regions.
The Agoufou site is located in the southern part of the Gourma region in Mali, which is a typical Sahelian grassland with 2 %–3 % tree cover (that was neglected in the simulation) on which occasional livestock grazing occurs. Correlation coefficients between the measured and modeled NEE and LAI over the period from 2007 to 2008 were 0.80 and 0.88, respectively. Nevertheless, the impact of drought on NEE in autumn 2007 was overestimated, probably because the simulated water content is lower than indicated by the measurements (Fig. 5a, bottom panel). We attribute this underestimation to a mismatch between the plot where initialization data were taken and the location of the measurements similar to what we observed at Nalohou, although not only the field capacity but also the wilting point is underestimated in this case. Some small deviations after a sudden LAI decrease in the middle of the first vegetation period can be seen (Fig. 5a, middle panel). This was most likely caused by a dry spell in September that triggered wilting of herbaceous vegetation but not of the grasses taken as representative vegetation here. On the one hand, this indicates that care has to be taken when selecting representative vegetation for a larger region; on the other hand, it shows that satellite data are of limited use when carrying out a comparison with estimates for specific sites. Belowground respiration also seems to be overestimated after the end of this vegetation period, which might be caused by the model assuming litter decomposition of grass material that was fully or partly removed by management activities. Overall, measurements indicate that the Agoufou site acted as a C sink of 890 kg C ha−1 yr−1, whereas the net C sequestration according to simulations was 761 kg C ha−1 yr−1.
At the Dahra site, which is in the northern part of Senegal, the footprint of the eddy-covariance tower is also largely dominated by grass vegetation with occasional trees, leading to a negligible tree cover of 3 % (Tagesson et al., 2015). There was general good agreement with NEE data (r=0.67), even though the simulations did not capture the peak net C uptake rates during the wet periods. Phenology also started too late, particularly in 2016 (Fig. 5b). The underestimation could be related to this site being more productive than average with respect to this region because of its relatively high nutrient availability (Tagesson et al., 2016). Nevertheless, both measurements and simulations indicate that Dahra was a substantial C sink for all years (2015–2017).
The Wankama-1 site in the southwest of the Republic of Niger is known as a fallow savanna (herbs and shrubs) where the land use remained unchanged from 2005 to 2012. The relatively high abundance of shrubs (Boulain et al., 2009) that are evergreen but have shading impacts on the surrounding grass cover is not represented in the model and leads to an overestimation of grass LAI during the vegetation period and an underestimation during the rest of the year. Correlation coefficients between measured and simulated NEE and LAI were 0.77 and 0.78, respectively. However, in some years such as 2006 and 2012, the model assumed longer vegetation periods and, thus, higher NEE which might be caused by a mismatch between physiological properties of assumed and actual species or an unusually intensive grazing that has not been adequately considered in the simulations (Fig. 5c). The average simulated NEE for Wankama-1 was an uptake of 1894 kg C ha−1 yr−1 (the measured value indicates 1505 kg C ha−1 yr−1 but major data gaps prevent the calculation of a meaningful annual average).
Regarding the Sudanian ecozone, the first core site is Sumbrungu, located in Ghana's Upper East Province. The overall correlation between NEE measurement and model simulations was high (r=0.89), even though simulations seemed to slightly overestimate the fluxes. According to the simulations, the maximum C uptake was in August and September at a rate of 29.3 kg C ha−1 d−1. The average C loss over the dry period varies considerably from 4320 kg C ha−1 (in 2013) to 1487 kg C ha−1 (in 2015), correlating strongly with the annual precipitation (679 mm of annual precipitation in 2013, whereas the normal precipitation amount is around 978 mm yr−1).
The second Sudanian site is Bontioli in the southwest of Burkina Faso. Here, absolute values of NEE were again overestimated by the model during the wet season. Nevertheless, simulations did capture the transition phase from dry and wet period (Fig. 5d), although the simulated vegetation period was about 2–3 weeks longer than indicated by the eddy-covariance measurements. However, there was a good correlation between measured and simulated LAI (r=0.95), possibly indicating that the efficiency of the plants in capturing C was underestimated. This might be caused by higher N availability than indicated by the deposition regime – for example, due to the deposition of fertile ash from nearby fires (Bauters et al., 2018).
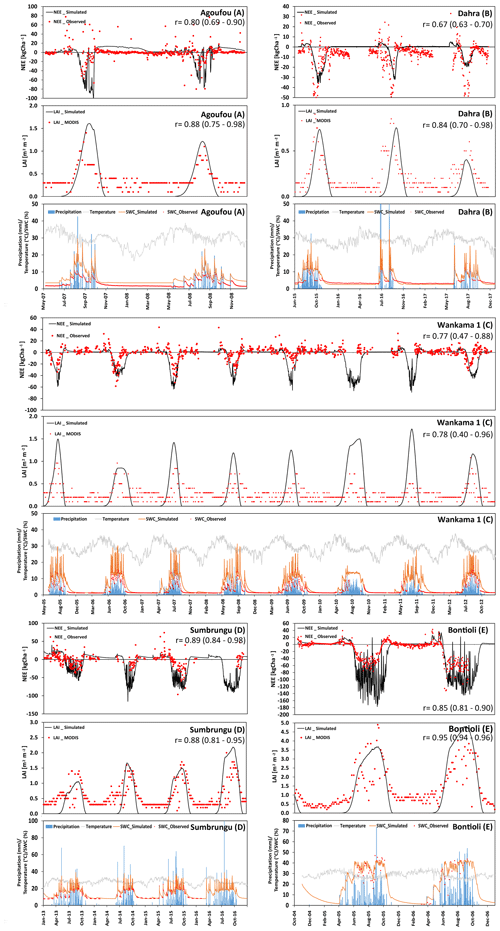
Figure 5(a–e) Time series of the observed and modeled NEE (kg ha−1; negative values represent a flux to the biosphere, and positive values represent a flux to the atmosphere), LAI (m2 m−2), and soil-water content (at 10 cm depth). In addition, precipitation and temperature at different grass-dominated sites in Sahelian zone are presented.
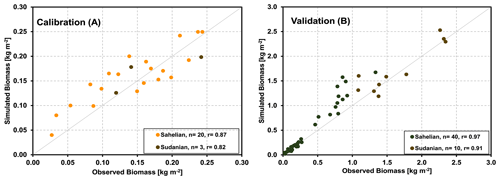
Figure 6Correlations between the observed and modeled biomass production at different grass-dominated sites in the Sahelian and Sudanian ecological zones for the calibration (a) and validation (b) data.
Similar to the core crop sites, we used the parameters obtained for the core sites for the validation of the model's ability to represent grassland savanna sites. In Fig. 6a, the aboveground biomass production simulated by the LandscapeDNDC model is compared with field observations of 23 samples from the five core sites. It demonstrates that the simulated aboveground biomass production generally agreed with the observations for all studied sites in the Sahelian and Sudanian ecological zones (correlation coefficient of 85 %). The validation exercise with additional 27 grass-dominated sites across Sahelian and Sudanian ecological zones is shown in Fig. 6b. It should be noted that some biomass yields in the validation sites are considerably larger than those found in any of the core sites.
3.3 Savanna mixed tree–grassland sites
We also investigated the NEE and LAI of three grassland sites with a considerable but different tree contribution. Two of these sites are located in the Sudanian zone (Nazinga and Bellefoungou) and one is located in the Sahelian zone (Kelma). For the Nazinga site in southern Burkina Faso the model was able to simulate the fluctuations in NEE and LAI over time series well (correlation coefficient of 0.79 and 0.85, respectively). However, the model underestimated ecosystem respiration during the dry period (especially in 2013), whereas LAI values (for both trees and grasses) were well simulated (Fig. 7a). The high variability in measured fluxes showed C releases during wet periods, indicating that some uncertainty exists with respect to species properties and the measured footprint, possibly involving other species during specific periods (Bliefernicht et al., 2018).
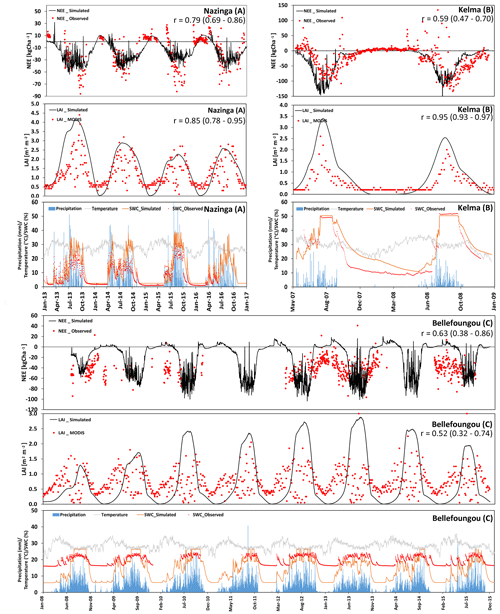
Figure 7(a–c) Time series of the observed and modeled NEE (kg ha−1; negative values represent a flux to the biosphere, and positive values represent a flux to atmosphere), LAI (m2 m−2), and soil-water content (at 10 cm depth). In addition, precipitation and temperature at different tree-dominated grassland/woodland sites in the Sahelian (Kelma) and Sudanian (Nazinga, Bellefoungou) ecological zones are presented.
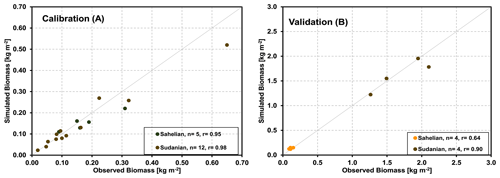
Figure 8Correlations between the observed and modeled biomass production at different tree-dominated grassland/woodland sites in Sahelian and Sudanian ecological zone through calibration and validation process.
The Kelma site, a facility located in the southern part of the Gourma region in Mali, is specific with respect to its water supply because it is a seasonally flooded open woodland. Patterns and magnitudes of NEE and LAI were generally well simulated by the model, although the onset of the vegetation period was estimated somewhat too early by the model (Fig. 7b). Furthermore, measurements indicated high levels of ecosystem respiration occurring at the end of the dry period in 2007, which could not be fully represented by the model. Such rewetting events are assumed to be related to increased decomposition and have been observed before (Epron et al., 2004; Grote et al., 2009b), but they remain challenging for soil C models to capture (see Fraser et al., 2016).
In contrast, LAI at Bellefoungou in the Djougou district, located in the northern part of Benin, was overestimated during the peak of the wet season and underestimated during transition phases (Fig. 7c), resulting in a relatively small correlation coefficient between the simulated and measured NEE and LAI (0.63 and 0.52, respectively). Again, this may be related to a relatively high density of (tree) species or to a mismatch between the selected tree and grass species and the actual species composition (Ago, 2016; Mamadou, 2014). On-site observations show that the different species renew or keep their leaves throughout the dry season, indicating that trees have access to deep ground water reservoirs, which is not considered in our simulations. Therefore, simulations cannot represent the continuous abundance of LAI and carbon sequestration throughout the dry season (Fig. 7c). It should also be noted that MODIS estimates are not very reliable at this site during the wet seasons because of fog formation which impacts infrared and visible remote sensing measurements. Nevertheless, the simulated NEE indicated a similar cumulative annual NEE budget of about −5660 kg C ha−1 yr−1 as compared to measurements.
As measured foliage or aboveground biomass data of trees were unavailable, we compare measured and simulated herbaceous biomass, as harvested at the peak of the vegetation period only (Fig. 8). For the five sites and across the two climate zones, simulations and measurements were highly correlated (calibration and validation correlation coefficients of 0.95 and 0.64 for the Sahelian and 0.98 and 0.90 for the Sudanian region, respectively).
Biogeochemical models can be used to determine terrestrial carbon pool developments and fluxes at temporal resolutions and regional scales that cannot be covered by field measurements. However, calibration and evaluation are needed for a range of representative sites in order to be reliable for assessing effects of environmental and anthropogenic (management) changes. Therefore, to date, this study presents the most extensive calibration and evaluation exercise of a biogeochemical model for natural and agricultural ecosystems in the Sahelian and Sudanian ecological zones of West Africa. More specifically, the LandscapeDNDC model framework was applied to 54 (12 core sites and 42 validation sites) intensive investigation sites where eddy-covariance and/or biomass measurements were available for more than 1 year. These sites covered the major natural vegetation types as well as the most important agricultural vegetation classes.
The results showed that the parameterized and complemented LandscapeDNDC model was able to represent C fluxes and pools for croplands, grasslands, and tree–grass mixtures in various combinations independent of nutrient availability and climatic conditions. Deviations from measurements could mostly be explained by simplifications in species initialization, which still only had a minor effect on overall carbon exchange and yield. Also, satellite observations (MODIS) had an overly coarse spatial resolution, meaning that they could not fully capture various parallel dynamics within one observation pixel. While C fluxes were mostly well represented by the model, soil C emission following rewetting of dry soils was occasionally underrepresented, which is a common problem in biogeochemical models. As the respective periods were relatively short and did not occur at all sites, the overall effect on the annual C balance was rather minor.
As the model only considers one representative plant species per natural ecosystem type, it is not able to account for species-specific combinations or a shift in species abundance that might change ecosystem sensitivity over longer periods. It is, however, feasible to run scenarios of land-cover and land-use changes that can happen rather quickly and investigate the impact on the regional carbon cycle considering an altered abundance of different ecosystems. Moreover, scenarios of environmental changes (e.g., changes in rainfall patterns, temperature, length of drought/rainy periods) that can develop over relatively short periods of time (and are thus faster than ecological adaptation responses) can be well investigated. With the evaluation presented here, the model is rendered particularly suitable as a decision support tool to explore the effect of climate-smart agricultural practices as well as various management practices in natural and agricultural land-use systems in West Africa on biosphere–atmosphere exchange. Furthermore, as the model is specifically designed to simulate trace gas exchanges (i.e., because of its detailed consideration of soil processes), greenhouse gas emissions from soils other than carbon releases can also be simulated (e.g., N2O and CH4). Thus, the model can be applied to assess further management effects such as different fertilization practices on West African semiarid ecosystems as well as to estimate the impacts on greenhouse gas emissions and on plant growth. However, it would be good if further evaluation with respect to such trace gas exchanges could be provided, particularly covering rewetting events.
The LandscapeDNDC model source code for released versions of the model is permanently available online at the Radar4KIT database (DOI: https://doi.org/10.35097/438; Butterbach-Bahl et al., 2021) along with all of the setup and model initialization information (DOI: https://doi.org/10.35097/437, Rahimi et al., 2021). It can be additionally downloaded upon request from the following KIT institutional website: https://ldndc.imk-ifu.kit.edu/download/download.php (last access: 16 June 2021). The version used in this study is “win64 ldndc-1.30.4”.
All data regarding soil properties, climate, and management that were used to initialize and run the model, as indicated in the text, are provided in the Radar4KIT database (DOI: https://doi.org/10.35097/437; Rahimi et al., 2021) and are freely downloadable. In addition, evaluation data were obtained from the FLUXNET (Pastorello et al., 2020; https://daac.ornl.gov/, last access: 16 June 2021), WASCAL Data Discovery Portal (Bliefernicht et al., 2018; https://wascal-dataportal.org/wascal_searchportal2/, last access: 16 June 2021), CARBOAFRICA project (Bombelli et al., 2009; https://www.carboafrica.eu/data_en.asp/, last access: 16 June 2021), and AMMA-CATCH database (Gallé and Feller, 2007; http://bd.amma-catch.org/, last access: 16 June 2021) websites with respect to gas exchange; and from the Land Processes Distributed Active Archive Center (LP DAAC) gateway (Myneni et al., 2015; https://lpdaac.usgs.gov/products/mcd15a3hv006/, last access: 16 June 2021) regarding LAI for all sites.
JR, RG, EH, and KBB conceived and designed the study. JR, RG, and DK preprocessed the data and parameterized and validated the model. EEA, AA, SB, JB, BC, AAD, UF, PH, TT, OR, and AKS provided all of the flux and biomass measurement data for the analysis. JR and RG wrote the first draft of the paper, and all co-authors contributed to further revisions.
The authors declare that they have no conflict of interest.
The authors appreciate the fruitful cooperation and data exchange with the EU-funded CASSECS project and the staff of the AMMA-CATCH observatory. Furthermore, some of the climate data were obtained from the NASA Langley Research Center (LaRC) POWER Project, funded through the NASA Earth Science/Applied Science Program. We acknowledge support by the KIT-Publication Fund of the Karlsruhe Institute of Technology.
This research has been supported by the UPSCALERS project (grant no. AURG II-1-074-2016), which is part of the
African Union Research Grant program financed through the agreement between the European Commission and the African Union Commission (grant no. DCI-PANAF/2015/307-078). We also acknowledge special funding supplied by the Swedish National Space Agency (SNSA).
The article processing charges for this open-access publication were covered by the Karlsruhe Institute of Technology (KIT).
This paper was edited by Hisashi Sato and reviewed by Zhipin Ai and one anonymous referee.
Ago, E. E.: Dynamique des flux de carbone entre l'atmosphère et des écosystèmes ouest-africains: cas des forêts et savanes sous climat soudanien au Bénin, Doctorat en Sciences Agronomiques et Ingénierie Biologique, Université de Liège, Gembloux, Belgique, 184 pp., 2016.
Ago, E. E., Agbossou, E. K., Galle, S., Cohard, J.-M., Heinesch, B., and Aubinet, M.: Long term observations of carbon dioxide exchange over cultivated savanna under a Sudanian climate in Benin (West Africa), Agric. Forest Meteorol., 197, 13–25, https://doi.org/10.1016/j.agrformet.2014.06.005, 2014.
Ago, E. E., Agbossou, E. K., Cohard, J.-M., Galle, S., and Aubinet, M.: Response of CO2 fluxes and productivity to water availability in two contrasting ecosystems in northern Benin (West Africa), Ann. For. Sci., 73, 483–500, https://doi.org/10.1007/s13595-016-0542-9, 2016.
Ahlström, A., Raupach, M. R., Schurgers, G., Smith, B., Arneth, A., Jung, M., Reichstein, M., Canadell, J. G., Friedlingstein, P., Jain, A. K., Kato, E., Poulter, B., Sitch, S., Stocker, B. D., Viovy, N., Wang, Y. P., Wiltshire, A., Zaehle, S., and Zeng, N.: The dominant role of semi-arid ecosystems in the trend and variability of the land CO2 sink, Science, 348, 895–899, https://doi.org/10.1126/science.aaa1668, 2015.
Ainsworth, E. A. and Rogers, A.: The response of photosynthesis and stomatal conductance to rising [CO2]: mechanisms and environmental interactions, Plant Cell Environ., 30, 258–270, https://doi.org/10.1111/j.1365-3040.2007.01641.x, 2007.
Akponikpè, P. B. I., Gérard, B., Michels, K., and Bielders, C.: Use of the APSIM model in long term simulation to support decision making regarding nitrogen management for pearl millet in the Sahel, European J. Agronomy, 32, 144–154, https://doi.org/10.1016/j.eja.2009.09.005, 2010.
Baldocchi, D. and Meyers, T.: On using eco-physiological, micrometeorological and biogeochemical theory to evaluate carbon dioxide, water vapor and trace gas fluxes over vegetation: a perspective, Agric. Forest Meteorol., 90, 1–25, https://doi.org/10.1016/S0168-1923(97)00072-5, 1998.
Baldocchi, D. and Xu, L.: Carbon exchange of deciduous broadleaved forests in temperate and Mediterranean regions, in: The Carbon Balance of Forest Biomes, edited by: Griffiths, H. and Jarvis, P. J., Garland Science/BIOS Scientific Publishers, London, 187–213, 2005.
Baldocchi, D. D., Xu, L., and Kiang, N.: How plant functional-type, weather, seasonal drought, and soil physical properties alter water and energy fluxes of an oak-grass savanna and an annual grassland, Agric. Forest Meteorol., 123, 13–39, https://doi.org/10.1016/j.agrformet.2003.11.006, 2004.
Ball, J. T., Woodrow, I. E., and Berry, J. A.: A model predicting stomatal conductance and its contribution to the control of photosynthesis under different environmental conditions, in: Prog. Photosyn. Res., edited by: Biggins, J., Martinus-Nijhoff Publishers, Dordrecht, the Netherlands, 221–224, 1987.
Batjes, N. H.: Harmonized soil profile data for applications at global and continental scales: updates to the WISE database, Soil Use Manage., 25, 124–127, https://doi.org/10.1111/j.1475-2743.2009.00202.x, 2008.
Baup, F., Mougin, E., de Rosnay, P., Timouk, F., and Chênerie, I.: Surface soil moisture estimation over the AMMA Sahelian site in Mali using ENVISAT/ASAR data, Remote. Sens. Environ., 109, 473–481, https://doi.org/10.1016/j.rse.2007.01.015, 2007.
Bauters, M., Drake, T. W., Verbeeck, H., Bodé, S., Hervé-Fernández, P., Zito, P., Podgorski, D. C., Boyemba, F., Makelele, I., Cizungu Ntaboba, L., Spencer, R. G. M., and Boeckx, P.: High fire-derived nitrogen deposition on central African forests, P. Natl. Acad. Sci. USA, 115, 549–554, https://doi.org/10.1073/pnas.1714597115, 2018.
Berger, S., Bliefernicht, J., Linstädter, A., Canak, K., Guug, S., Heinzeller, D., Hingerl, L., Mauder, M., Neidl, F., Quansah, E., Salack, S., Steinbrecher, R., and Kunstmann, H.: The impact of rain events on CO2 emissions from contrasting land use systems in semi-arid West African savannas, Sci. Total Environ., 647, 1478–1489, https://doi.org/10.1016/j.scitotenv.2018.07.397, 2019.
Bernacchi, C. J., Portis, A. R., Nakano, H., von Caemmerer, S., and Long, S. P.: Temperature Response of Mesophyll Conductance. Implications for the Determination of Rubisco Enzyme Kinetics and for Limitations to Photosynthesis in Vivo, Plant Physiol., 130, 1992–1998, https://doi.org/10.1104/pp.008250, 2002.
Bliefernicht, J., Berger, S., Salack, S., Guug, S., Hingerl, L., Heinzeller, D., Mauder, M., Steinbrecher, R., Steup, G., Bossa, A. Y., Waongo, M., Quansah, E., Balogun, A. A., Yira, Y., Arnault, J., Wagner, S., Klein, C., Gessner, U., Knauer, K., Straub, A., Schönrock, R., Kunkel, R., Okogbue, E. C., Rogmann, A., Neidl, F., Jahn, C., Diekkrüger, B., Aduna, A., Barry, B., and Kunstmann, H.: The WASCAL Hydrometeorological Observatory in the Sudan Savanna of Burkina Faso and Ghana, Vadose Zone J., 17, 180065, https://doi.org/10.2136/vzj2018.03.0065, 2018.
Boke-Olén, N., Lehsten, V., Ardö, J., Beringer, J., Eklundh, L., Holst, T., Veenendaal, E., and Tagesson, T.: Estimating and Analyzing Savannah Phenology with a Lagged Time Series Model, PLoS ONE, 11, e0154615–e0154615, https://doi.org/10.1371/journal.pone.0154615, 2016.
Bocksberger, G., Schnitzler, J., Chatelain, C., Daget, P., Janssen, T., Schmidt, M., Thiombiano, A., and Zizka, G.: Climate and the distribution of grasses in West Africa, J. Veg. Sci., 27, 306–317, https://doi.org/10.1111/jvs.12360, 2016.
Bombelli, A., Henry, M., Castaldi, S., Adu-Bredu, S., Arneth, A., de Grandcourt, A., Grieco, E., Kutsch, W. L., Lehsten, V., Rasile, A., Reichstein, M., Tansey, K., Weber, U., and Valentini, R.: An outlook on the Sub-Saharan Africa carbon balance, Biogeosciences, 6, 2193–2205, https://doi.org/10.5194/bg-6-2193-2009, 2009.
Boone, R. B., Galvin, K. A., Coughenour, M. B., Hudson, J. W., Weisberg, P. J., Vogel, C. H., and Ellis, J. E.: Ecosystem modeling adds value to a south african climate forecast, Clim. Change, 64, 317–340, https://doi.org/10.1023/B:CLIM.0000025750.09629.48, 2004.
Boulain, N., Cappelaere, B., Ramier, D., Issoufou, H. B. A., Halilou, O., Seghieri, J., Guillemin, F., Oï, M., Gignoux, J., and Timouk, F.: Towards an understanding of coupled physical and biological processes in the cultivated Sahel – 2. Vegetation and carbon dynamics, J. Hydrol., 375, 190–203, https://doi.org/10.1016/j.jhydrol.2008.11.045, 2009.
Boyd, R. A., Gandin, A., and Cousins, A. B.: Temperature Responses of C4 Photosynthesis: Biochemical Analysis of Rubisco, Phosphoenolpyruvate Carboxylase, and Carbonic Anhydrase in Setaria viridis, Plant Physiol., 169, 1850–1861, https://doi.org/10.1104/pp.15.00586, 2015.
Brümmer, C., Falk, U., Papen, H., Szarzynski, J., Wassmann, R., and Brüggemann, N.: Diurnal, seasonal, and interannual variation in carbon dioxide and energy exchange in shrub savanna in Burkina Faso (West Africa), J. Geophys. Res., 113, G02030, https://doi.org/10.1029/2007JG000583, 2008.
Buba, T.: Prediction equations for estimating tree height, crown diameter, crown height and crown ratio of Parkia biglobosa in the Nigerian guinea savanna, Afr. J. Agr. Res., 7, 6541–6543, https://doi.org/10.5897/AJAR12.276, 2013.
Buchhorn, M., Smets, B., Bertels, L., Lesiv, M., Tsendbazar, N.-E., Masiliunas, D., Linlin, L., Herold, M., and Fritz, S.: Copernicus Global Land Service: Land Cover 100m: Collection 3: epoch 2019: Globe (Version V3.0.1) [Data set], Zenodo, https://doi.org/10.5281/zenodo.3939050, 2020.
Butterbach-Bahl, K., Grote, R., Haas, E., Kiese, R., Klatt, S., and Kraus, D.: LandscapeDNDC (v1.30.4) [code], Karlsruhe Institute of Technology (KIT), https://doi.org/10.35097/438, 2021.
Caldararu, S., Purves, D. W., and Smith, M. J.: The impacts of data constraints on the predictive performance of a general process-based crop model (PeakN-crop v1.0), Geosci. Model Dev., 10, 1679–1701, https://doi.org/10.5194/gmd-10-1679-2017, 2017.
Camargo, A. P., Marin, F. R., Sentelhas, P. C., and Picini, A. G.: Adjust of the Thornthwaite's method to estimate the potential evapotranspiration for arid and superhumid climates, based on daily temperature amplitude, Bras. Agrometeorol., 7, 251–257, https://doi.org/10.1007/s00704-019-02873-1, 1999.
Cappelaere, B., Descroix, L., Lebel, T., Boulain, N., Ramier, D., Laurent, J. P., Favreau, G., Boubkraoui, S., Boucher, M., Bouzou Moussa, I., Chaffard, V., Hiernaux, P., Issoufou, H. B. A., Le Breton, E., Mamadou, I., Nazoumou, Y., Oi, M., Ottlé, C., and Quantin, G.: The AMMA-CATCH experiment in the cultivated Sahelian area of south-west Niger – Investigating water cycle response to a fluctuating climate and changing environment, J. Hydrol., 375, 34–51, https://doi.org/10.1016/j.jhydrol.2009.06.021, 2009.
Chandra, A. and Dubey, A.: Evaluation of genus Cenchrus based on malondialdehyde, proline content, specific leaf area and carbon isotope discrimination for drought tolerance and divergence of species at DNA level, Acta Physiol. Plant., 30, 53–61, https://doi.org/10.1007/s11738-007-0090-x, 2008.
Chen, Q., Baldocchi, D., Gong, P., and Dawson, T.: Modeling radiation and photosynthesis of a heterogeneous savanna woodland landscape with a hierarchy of model complexities, Agric. Forest Meteorol., 148, 1005–1020, https://doi.org/10.1016/j.agrformet.2008.01.020, 2008.
Collatz, G. J., Ribas-Carbo, M., and Berry, J. A.: Coupled photosynthesis-stomatal conductance model for leaves of C4 plants, Aust. J. Plant Physiol., 19, 519–538, https://doi.org/10.1071/PP9920519 1992.
Dagbenonbakin, G. D.: Productivity and water use efficiency of important crops in the upper Oueme Catchment: influence of nutrient limitations, nutrient balances and soil fertility, PhD, Hohe Landwirtschaftliche Fakultät, Rheinischen Friedrich-Wilhelms-Universität Bonn, Bonn, 212 pp., 2005.
Da Matta, F. M., Loos, R. A., Rodrigues, R., and Barros, R.: Actual and potential photosynthetic rates of tropical crop species, Revista Brasileira de Fisiologia Vegetal, 13, 24–32, https://doi.org/10.1590/S0103-31312001000100003., 2001.
Dayamba, S. D., Djoudi, H., Zida, M., Sawadogo, L., and Verchot, L.: Biodiversity and carbon stocks in different land use types in the Sudanian Zone of Burkina Faso, West Africa, Agr. Ecosyst. Environ., 216, 61–72, https://doi.org/10.1016/j.agee.2015.09.023, 2016.
de Jong, S. M. and Jetten, V. G.: Estimating spatial patterns of rainfall interception from remotely sensed vegetation indices and spectral mixture analysis, Int. J. Geogr. Inf. Sci., 21, 529–545, https://doi.org/10.1080/13658810601064884, 2007.
de Rosnay, P., Gruhier, C., Timouk, F., Baup, F., Mougin, E., Hiernaux, P., Kergoat, L., and LeDantec, V.: Multi-scale soil moisture measurements at the Gourma meso-scale site in Mali, J. Hydrol., 375, 241–252, https://doi.org/10.1016/j.jhydrol.2009.01.015, 2009.
Delon, C., Mougin, E., Serça, D., Grippa, M., Hiernaux, P., Diawara, M., Galy-Lacaux, C., and Kergoat, L.: Modelling the effect of soil moisture and organic matter degradation on biogenic NO emissions from soils in Sahel rangeland (Mali), Biogeosciences, 12, 3253–3272, https://doi.org/10.5194/bg-12-3253-2015, 2015.
Delon, C., Galy-Lacaux, C., Serça, D., Personne, E., Mougin, E., Adon, M., Le Dantec, V., Loubet, B., Fensholt, R., and Tagesson, T.: Modelling land–atmosphere daily exchanges of NO, NH3, and CO2 in a semi-arid grazed ecosystem in Senegal, Biogeosciences, 16, 2049–2077, https://doi.org/10.5194/bg-16-2049-2019, 2019.
de Souza Nóia Júnior, R., Amaral, G. C., Pezzopane, J. E. M., Fonseca, M. D. S., Câmara da Silva, A. P., and Xavier, T. M. T.: Ecophysiological acclimatization to cyclic water stress in Eucalyptus, J. Forestry Res., 31, 797–806, https://doi.org/10.1007/s11676-019-00926-9, 2020.
Dimobe, K., Kouakou, J. L. N. D., Tondoh, J. E., Zoungrana, B. J.-B., Forkuor, G., and Ouédraogo, K.: Predicting the Potential Impact of Climate Change on Carbon Stock in Semi-Arid West African Savannas, Land, 7, 124, https://doi.org/10.3390/land7040124, 2018.
Diouf, A. A., Brandt, M., Verger, A., Jarroudi, M. E., Djaby, B., Fensholt, R., Ndione, J. A., and Tychon, B.: Fodder Biomass Monitoring in Sahelian Rangelands Using Phenological Metrics from FAPAR Time Series, Remote Sens., 7, 9122–9148, https://doi.org/10.3390/rs70709122, 2015.
Dirnböck, T., Kraus, D., Grote, R., Klatt, S., Kobler, J., Schindlbacher, A., Seidl, R., Thom, D., and Kiese, R.: Substantial understory contribution to the C sink of a European temperate mountain forest landscape, Landscape Ecol., 35, 483–499, https://doi.org/10.1007/s10980-019-00960-2 2020.
Do, F. C., Goudiaby, V. A., Gimenez, O., Diagne, A. L., Diouf, M., Rocheteu, A., and Akpo, L. E.: Environmental influence on canopy phenology in the dry tropics, Forest Ecol. Manage., 215, 319–328, https://doi.org/10.1016/j.foreco.2005.05.022, 2005.
Elberling, B., Fensholt, R., Larsen, L., Petersen, A.-I. S., and Sandholt, I.: Water content and land use history controlling soil CO2 respiration and carbon stock in savanna soil and groundnut fields in semi-arid Senegal, Danish J. Geogr., 103, 47–56, https://doi.org/10.1080/00167223.2003.10649491, 2003.
Epron, D., Nouvellon, Y., Roupsard, O., Mouvondy, W., Mabiala, A., Saint-Andre, L., Joffre, R., Jourdan, C., Bonnefond, J. M., Berbigier, P., and Hamel, O.: Spatial and temporal variations of soil respiration in a Eucalyptus plantation in Congo, Forest Ecol. Manage., 202, 149–160, https://doi.org/10.1016/j.foreco.2004.07.019, 2004.
Farquhar, G. D., Von Caemmerer, S., and Berry, J. A.: A biochemical model of photosynthetic CO2 assimilation in leaves of C3 species, Planta, 149, 78–90, https://doi.org/10.1007/BF00386231, 1980.
Faye, B., Webber, H., Naab, J. B., MacCarthy, D. S., Adam, M., Ewert, F., Lamers, J. P. A., Schleussner, C.-F., Ruane, A., Gessner, U., Hoogenboom, G., Boote, K., Shelia, V., Saeed, F., Wisser, D., Hadir, S., Laux, P., and Gaiser, T.: Impacts of 1.5 versus 2.0 ∘C on cereal yields in the West African Sudan Savanna, Environ. Res. Lett., 13, 034014, https://doi.org/10.1088/1748-9326/aaab40, 2018.
February, E. and Higgins, S.: The distribution of tree and grass roots in savannas in relation to soil nitrogen and water, S. Afr. J. Bot., 76, 517–523, https://doi.org/10.1016/j.sajb.2010.04.001, 2010.
Feng, X. and Dietze, M.: Scale dependence in the effects of leaf ecophysiological traits on photosynthesis: Bayesian parameterization of photosynthesis models, New Phytol., 200, 1132–1144, https://doi.org/10.1111/nph.12454, 2013.
Fraser, F. C., Corstanje, R., Deeks, L. K., Harris, J. A., Pawlett, M., Todman, L. C., Whitmore, A. P., and Ritz, K.: On the origin of carbon dioxide released from rewetted soils, Soil Biology and Biochemistry, 101, 1–5, https://doi.org/10.1016/j.soilbio.2016.06.032, 2016.
Fürstenau Togashi, H., Prentice, I. C., Atkin, O. K., Macfarlane, C., Prober, S. M., Bloomfield, K. J., and Evans, B. J.: Thermal acclimation of leaf photosynthetic traits in an evergreen woodland, consistent with the coordination hypothesis, Biogeosciences, 15, 3461–3474, https://doi.org/10.5194/bg-15-3461-2018, 2018.
Gallé, A. and Feller, U.: Changes of photosynthetic traits in beech saplings (Fagus sylvatica) under severe drought stress and during recovery, Physiol. Plant., 131, 412–421, 2007.
Galy-Lacaux, C., Delon, C., Solmon, F., Adon, M., Yoboué, V., Mphepya, J., Pienaar, J. J., Diop, B., Sigha, L., Dungall, L., Akpo, A., Mougin, E., Gardrat, E., and Castera, P.: Dry and Wet Atmospheric Nitrogen Deposition in West Central Africa, in: Nitrogen Deposition, Critical Loads and Biodiversity, edited by: Sutton, M., Mason, K., Sheppard, L., Sverdrup, H., Haeuber, R., and Hicks, W., Springer, Dordrecht, 2014.
Gash, J. H. C., Lloyd, C. R., and Lachaud, G.: Estimating sparse forest rainfall interception with an analytical model, J. Hydrol., 170, 79–86, https://doi.org/10.1016/0022-1694(95)02697-N, 1995.
Geerling, C.: The status of the woody species of the Sudan and Sahel zones of West Africa, Forest Ecol. Manage., 13, 247–255, https://doi.org/10.1016/0378-1127(85)90038-6, 1985.
Gessner, U., Niklaus, M., Kuenzer, C., and Dech, S.: Intercomparison of Leaf Area Index Products for a Gradient of Sub-Humid to Arid Environments in West Africa, Remote Sens., 5, 1235–1257, https://doi.org/10.3390/rs5031235, 2013.
Gleason, S. M., Wiggans, D. R., Bliss, C. A., Comas, L. H., Cooper, M., DeJonge, K. C., Young, J. S., and Zhang, H.: Coordinated decline in photosynthesis and hydraulic conductance during drought stress in Zea mays, Flora, 227, 1–9, https://doi.org/10.1016/j.flora.2016.11.017, 2017.
Groenendijk, M., Dolman, A. J., van der Molen, M. K., Leuning, R., Arneth, A., Delpierre, N., Gash, J. H. C., Lindroth, A., Richardson, A. D., Verbeeck, H., and Wohlfahrt, G.: Assessing parameter variability in a photosynthesis model within and between plant functional types using global Fluxnet eddy covariance data, Agric. Forest Meteorol., 151, 22–38, https://doi.org/10.1016/j.agrformet.2010.08.013, 2011.
Grossiord, C., Sevanto, S., Adams, H. D., Collins, A. D., Dickman, L. T., McBranch, N., Michaletz, S. T., Stockton, E. A., Vigil, M., and McDowell, N. G.: Precipitation, not air temperature, drives functional responses of trees in semi-arid ecosystems, J. Ecol., 105, 163–175, https://doi.org/10.1111/1365-2745.12662, 2017.
Grote, R.: Integrating dynamic morphological properties into forest growth modeling. II. Allocation and mortality, Forest Ecol. Manage., 111, 193–210, https://doi.org/10.1016/S0378-1127(98)00328-4, 1998.
Grote, R.: Sensitivity of volatile monoterpene emission to changes in canopy structure – A model based exercise with a process-based emission model, New Phytol., 173, 550–561, https://doi.org/10.1111/j.1469-8137.2006.01946.x, 2007.
Grote, R., Lavoir, A. V., Rambal, S., Staudt, M., Zimmer, I., and Schnitzler, J.-P.: Modelling the drought impact on monoterpene fluxes from an evergreen Mediterranean forest canopy, Oecologia, 160, 213–223, https://doi.org/10.1007/s00442-009-1298-9, 2009a.
Grote, R., Lehmann, E., Brümmer, C., Brüggemann, N., Szarzynski, J., and Kunstmann, H.: Modelling and observation of biosphere-atmosphere interactions in natural savannah in Burkina Faso, West Africa, Phys. Chem. Earth, 34, 251–260, https://doi.org/10.1016/j.pce.2008.05.003, 2009b.
Grote, R., Kiese, R., Grünwald, T., Ourcival, J.-M., and Granier, A.: Modelling forest carbon balances considering tree mortality and removal, Agric. Forest Meteorol., 151, 179–190, https://doi.org/10.1016/j.agrformet.2010.10.002, 2011a.
Grote, R., Korhonen, J., and Mammarella, I.: Challenges for evaluating process-based models of gas exchange at forest sites with fetches of various species, For. Syst., 20, 389–406, https://doi.org/10.5424/fs/20112003-11084, 2011b.
Grote, R., Kraus, D., Weis, W., Ettl, R., and Göttlein, A.: Dynamic coupling of allometric ratios to a process-based forest growth model for estimating the impacts of stand density changes, Forestry, 93, 601–615, https://doi.org/10.1093/forestry/cpaa002, 2020.
Guenther, A., Otter, L., Zimmerman, P., Greenberg, J., Scholes, R., and Scholes, M.: Biogenic hydrocarbon emissions from southern Africa savannas, J. Geophys. Res., 101, 25859–25865, https://doi.org/10.1029/96JD02597, 1996.
Haas, E., Klatt, S., Fröhlich, A., Werner, C., Kiese, R., Grote, R., and Butterbach-Bahl, K.: LandscapeDNDC: A process model for simulation of biosphere-atmosphere-hydrosphere exchange processes at site and regional scale, Landscape Ecol., 28, 615–636, https://doi.org/10.1007/s10980-012-9772-x, 2013.
Hartley, A. J., Parker, D. J., Garcia-Carreras, L., and Webster, S.: Simulation of vegetation feedbacks on local and regional scale precipitation in West Africa, Agric. Forest Meteorol., 222, 59–70, https://doi.org/10.1016/j.agrformet.2016.03.001, 2016.
Hiernaux, P. and Ayantunde, A. A.: The Fakara: a semi-arid agro-ecosystem under stress. Report of research activities, first phase (July 2002–June 2004) of the DMP-GEF Program ILRI, Nairobi (Kenya), GEF/2711-02-4516, 95, 2004.
Hiernaux, P., Mougin, E., Diarra, L., Soumaguel, N., Lavenu, F., Tracol, Y., and Diawara, M. O.: Sahelian rangeland response to changes in rainfall over two decades in the Gourma region, Mali, J. Hydrol., 375, 114–127, https://doi.org/10.1016/j.jhydrol.2008.11.005, 2009.
Holá, D., Benešová, M., Honnerová, J., Hnilička, F., Rothová, O., Kočová, M., and Hniličková, H.: The evaluation of photosynthetic parameters in maize inbred lines subjected to water deficiency: Can these parameters be used for the prediction of performance of hybrid progeny?, Photosynthetica, 48, 545–558, https://doi.org/10.1007/s11099-010-0072-x, 2010.
Ivanov, V. Y., Bras, R. L., and Vivoni, E. R.: Vegetation-hydrology dynamics in complex terrain of semiarid areas: 1. A mechanistic approach to modeling dynamic feedbacks, Water Resour. Res., 44, W03429, https://doi.org/10.1029/2006WR005588, 2008.
Jolly, W. M. and Running, S. W.: Effects of precipitation and soil water potential on drought deciduous phenology in the Kalahari, Glob. Change Biol., 10, 303–308, https://doi.org/10.1046/j.1365-2486.2003.00701.x, 2004.
Kahiu, M. N. and Hanan, N. P.: Estimation of Woody and Herbaceous Leaf Area Index in Sub-Saharan Africa Using MODIS Data, J. Geophys. Res.-Biogeo., 123, 3–17, https://doi.org/10.1002/2017JG004105, 2018.
Kalariya, K. A., Singh, A. L., Goswami, N., Mehta, D., Mahatma, M. K., Ajay, B. C., Chakraborty, K., Zala, P. V., Chaudhary, V., and Patel, C. B.: Photosynthetic characteristics of peanut genotypes under excess and deficit irrigation during summer, Physiol. Mol. Biol. Plants, 21, 317–327, https://doi.org/10.1007/s12298-015-0300-8, 2015.
Kaptue Tchuente, A. T., Roujean, J. L., and Faroux, S.: ECOCLIMAP-II: An ecosystem classification and land surface parameters database of Western Africa at 1 km resolution for the African Monsoon Multidisciplinary Analysis (AMMA) project, Remote Sens. Environ., 114, 961–976, https://doi.org/10.1016/j.rse.2009.12.008, 2010.
Kattge, J. and Knorr, W.: Temperature acclimation in a biochemical model of photosynthesis: a reanalysis of data from 36 species, Plant Cell Environ., 30, 1176–1190, https://doi.org/10.1111/j.1365-3040.2007.01690.x, 2007.
Kebbas, S., Lutts, S., and Aid, F.: Effect of drought stress on the photosynthesis of Acacia tortilis subsp. raddiana at the young seedling stage, Photosynthetica, 53, 288–298, https://doi.org/10.1007/s11099-015-0113-6, 2015.
Ker, A.: Farming Systems of the African Savanna, International Development Research Centre, Ottawa, Canada, 166 pp., 1995.
Kgope, B. S. and Musil, C. F.: Differential photosynthetic responses of broad- and fine-leafed savanna trees to elevated temperatures, S. Afr. J. Bot., 70, 760–766, https://doi.org/10.1016/S0254-6299(15)30177-0, 2004.
Kiese, R., Heinzeller, C., Werner, C., Wochele, S., Grote, R., and Butterbach-Bahl, K.: Quantification of nitrate leaching from German forest ecosystems by use of a process oriented biogeochemical model, Environ. Pollut., 159, 3204–3214, https://doi.org/10.1016/j.envpol.2011.05.004, 2011.
Kim, J. and Verma, S. B.: Modeling canopy photosynthesis: scaling up from a leaf to canopy in a temperate grassland ecosystem, Agric. Forest Meteorol., 57, 187–208, https://doi.org/10.1016/0168-1923(91)90086-6, 1991.
Knauer, J., Werner, C., and Zaehle, S.: Evaluating stomatal models and their atmospheric drought response in a land surface scheme: A multibiome analysis, J. Geophys. Res.-Biogeo., 120, 1894–1911, https://doi.org/10.1002/2015jg003114, 2015.
Körner, C., Scheel, J., and Bauer, H.: Maximum leaf diffusive conductance in vascular plants, Photosynthetica, 13, 45–82, 1979.
Kothavala, Z., Arain, M. A., Black, T. A., and Verseghy, D.: The simulation of energy, water vapor and carbon dioxide fluxes over common crops by the Canadian Land Surface Scheme (CLASS), Agric. Forest Meteorol., 133, 89–108, https://doi.org/10.1016/j.agrformet.2005.08.007, 2005.
Kraus, D., Weller, S., Klatt, S., Haas, E., Wassmann, R., Kiese, R., and Butterbach-Bahl, K.: A new LandscapeDNDC biogeochemical module to predict CH4 and N2O emissions from lowland rice and upland cropping systems, Plant Soil, 386, 125–149, https://doi.org/10.1007/s11104-014-2255-x, 2015.
Kraus, D., Weller, S., Klatt, S., Santabárbara, I., Haas, E., Wassmann, R., Werner, C., Kiese, R., and Butterbach-Bahl, K.: How well can we assess impacts of agricultural land management changes on the total greenhouse gas balance (CO2, CH4 and N2O) of tropical rice-cropping systems with a biogeochemical model?, Agr. Ecosyst. Environ., 224, 104–115, https://doi.org/10.1016/j.agee.2016.03.037, 2016.
Kucharik, C. J., Barford, C. C., El Maayar, M., Wofsy, S. C., Monson, R. K., and Baldocchi, D. D.: A multiyear evaluation of a Dynamic Global Vegetation Model at three AmeriFlux forest sites: Vegetation structure, phenology, soil temperature, and CO2 and H2O vapor exchange, Ecol. Modelling, 196, 1–31, https://doi.org/10.1016/j.ecolmodel.2005.11.031, 2006.
Leuning, R.: A critical appraisal of a combined stomatal-photosynthesis model for C3 plants, Plant Cell Environ., 18, 339–355, https://doi.org/10.1111/j.1365-3040.1995.tb00370.x, 1995.
Li, C., Frolking, S., and Frolking, T. A.: A model of nitrous oxide evolution from soil driven by rainfall events: 1. Model structure and Sensitivity, J. Geophys. Res., 97, 9759–9776, https://doi.org/10.1029/92JD00509, 1992.
Liebermann, R., Breuer, L., Houska, T., Kraus, D., Moser, G., and Kraft, P.: Simulating Long-Term Development of Greenhouse Gas Emissions, Plant Biomass, and Soil Moisture of a Temperate Grassland Ecosystem under Elevated Atmospheric CO2, Agronomy, 10, 50, https://doi.org/10.3390/agronomy10010050, 2020.
Lindauer, M., Schmid, H. P., Grote, R., Mauder, M., Steinbrecher, R., and Wolpert, B.: Net ecosystem exchange over a non-cleared wind-throw-disturbed upland spruce forest – measurements and simulations Agric. Forest Meteorol., 197, 219–234, https://doi.org/10.1016/j.agrformet.2014.07.005, 2014.
Livesley, S. J., Grover, S., Hutley, L. B., Jamali, H., Butterbach-Bahl, K., Fest, B., Beringer, J., and Arndt, S. K.: Seasonal variation and fire effects on CH4, N2O and CO2 exchange in savanna soils of northern Australia, Agric. Forest Meteorol., 151, 1440–1452, https://doi.org/10.1016/j.agrformet.2011.02.001, 2011.
Loustau, D., Berbigier, P., Granier, A., and Moussa, F. E. H.: Interception loss, throughfall and stemflow in a maritime pine stand. I. Variability of throughfall and stemflow beneath the pine canopy, J. Hydrol., 138, 449–467, https://doi.org/10.1016/0022-1694(92)90130-N, 1992.
Mamadou, O.: Etude des flux d'Evapotranspiration en climat soudanien: comportement comparé de deux couverts végétaux au Bénin, Université de Grenoble (France) et Université d'Abomey-Calavi (Bénin), Abomey-Calavi, 2014.
Martin, M. J., Stirling, C. M., Humphries, S. W., and Long, S. P.: A process-based model to predict the effects of climatic change on leaf isoprene emission rates, Ecol. Model., 131, 161–174, https://doi.org/10.1016/S0304-3800(00)00258-1, 2000.
Massad, R.-S., Tuzet, A., and Bethenod, O.: The effect of temperature on C4-type leaf photosynthesis parameters, Plant Cell Environ., 30, 1191–1204, https://doi.org/10.1111/j.1365-3040.2007.01691.x, 2007.
Massad, R. S., Lathière, J., Strada, S., Perrin, M., Personne, E., Stéfanon, M., Stella, P., Szopa, S., and de Noblet-Ducoudré, N.: Reviews and syntheses: influences of landscape structure and land uses on local to regional climate and air quality, Biogeosciences, 16, 2369–2408, https://doi.org/10.5194/bg-16-2369-2019, 2019.
Merbold, L., Ardö, J., Arneth, A., Scholes, R. J., Nouvellon, Y., de Grandcourt, A., Archibald, S., Bonnefond, J. M., Boulain, N., Brueggemann, N., Bruemmer, C., Cappelaere, B., Ceschia, E., El-Khidir, H. A. M., El-Tahir, B. A., Falk, U., Lloyd, J., Kergoat, L., Le Dantec, V., Mougin, E., Muchinda, M., Mukelabai, M. M., Ramier, D., Roupsard, O., Timouk, F., Veenendaal, E. M., and Kutsch, W. L.: Precipitation as driver of carbon fluxes in 11 African ecosystems, Biogeosciences, 6, 1027–1041, https://doi.org/10.5194/bg-6-1027-2009, 2009.
Mougin, E., Lo Seena, D., Rambal, S., Gaston, A., and Hiernaux, P.: A regional Sahelian grassland model to be coupled with multispectral satellite data. I: Model description and validation, Remote. Sens. Environ., 52, 181–193, https://doi.org/10.1016/0034-4257(94)00126-8, 1995.
Mougin, E., Hiernaux, P., Kergoat, L., Grippa, M., de Rosnay, P., Timouk, F., Le Dantec, V., Demarez, V., Lavenu, F., Arjounin, M., Lebel, T., Soumaguel, N., Ceschia, E., Mougenot, B., Baup, F., Frappart, F., Frison, P. L., Gardelle, J., Gruhier, C., Jarlan, L., Mangiarotti, S., Sanou, B., Tracol, Y., Guichard, F., Trichon, V., Diarra, L., Soumaré, A., Koité, M., Dembélé, F., Lloyd, C., Hanan, N. P., Damesin, C., Delon, C., Serça, D., Galy-Lacaux, C., Seghieri, J., Becerra, S., Dia, H., Gangneron, F., and Mazzega, P.: The AMMA-CATCH Gourma observatory site in Mali: Relating climatic variations to changes in vegetation, surface hydrology, fluxes and natural resources, J. Hydrol., 375, 14–33, https://doi.org/10.1016/j.jhydrol.2009.06.045, 2009.
Mougin, E., Diawara, M. O., Soumaguel, N., Maïga, A. A., Demarez, V., Hiernaux, P., Grippa, M., Chaffard, V., and Ba, A.: A leaf area index data set acquired in Sahelian rangelands of Gourma in Mali over the 2005–2017 period, Earth Syst. Sci. Data, 11, 675–686, https://doi.org/10.5194/essd-11-675-2019, 2019.
Myneni, R., Knyazikhin, Y., and Park, T.: MCD15A3H MODIS/Terra+Aqua Leaf Area Index/FPAR 4-day L4 Global 500 m SIN Grid V006 [data set], NASA EOSDIS Land Processes DAAC, https://doi.org/10.5067/MODIS/MCD15A3H.006 (last access: 16 June 2021), 2015.
Odekunle, T. O., Andrew, O., and Aremu, S. O.: Towards a wetter Sudano-Sahelian ecological zone in twenty-first century Nigeria, Weather, 63, 66–70, https://doi.org/10.1002/wea.172, 2008.
Pallas, J. E. and Samish, Y. B.: Photosynthetic response of peanut, Crop Sci., 14, 478–482, https://doi.org/10.2135/cropsci1974.0011183X001400030042x, 1974.
Pastorello, G., Trotta, C., Canfora, E., Chu, H., Christianson, D., Cheah, Y.-W., Poindexter, C., Chen, J., Elbashandy, A., Humphrey, M., Isaac, P., Polidori, D., Ribeca, A., Ingen, C., Zhang, L., Amiro, B., Ammann, C., Arain, M., and Ardö, J.: The FLUXNET2015 dataset and the ONEFlux processing pipeline for eddy covariance data, Sci. Data, 7, 225, https://doi.org/10.1038/s41597-020-0534-3, 2020.
Pereira, A. R. and Pruitt, W. O.: Adaptation of the Thornthwaite scheme for estimating daily reference evapotranspiration, Agric. Water Manage., 66, 251–257, https://doi.org/10.1016/j.agwat.2003.11.003, 2004.
Pielke, R. A., Pitman, A., Niyogi, D., Mahmood, R., McAlpine, C., Hossain, F., Goldewijk, K. K., Nair, U., Betts, R., Fall, S., Reichstein, M., Kabat, P., and de Noblet, N.: Land use/land cover changes and climate: modeling analysis and observational evidence, Wiley Interdisciplinary Reviews: Climate Change, 2, 828–850, https://doi.org/10.1002/wcc.144, 2011.
Pitman, A. J.: The evolution of, and revolution in, land surface schemes designed for climate models, Int. J. Climatol., 23, 479–510, https://doi.org/10.1002/joc.893, 2003.
Quansah, E., Mauder, M., Balogun, A. A., Amekudzi, L. K., Hingerl, L., Bliefernicht, J., and Kunstmann, H.: Carbon dioxide fluxes from contrasting ecosystems in the Sudanian Savanna in West Africa, Carbon Balance Manag., 10, 1–17, https://doi.org/10.1186/s13021-014-0011-4, 2015.
Quenum, G. M. L. D., Klutse, N. A. B., Dieng, D., Laux, P., Arnault, J., Kodja, J. D., and Oguntunde, P. G.: Identification of Potential Drought Areas in West Africa Under Climate Change and Variability, Earth Syst. Environ., 3, 429–444, https://doi.org/10.1007/s41748-019-00133-w, 2019.
Rahimi, J., Ago, E. E., Ayantunde, A., Bogaert, J., Butterbach-Bahl, K., Cappelaere, B., Demarty, J., Diouf, A. A., Falk, U., Haas, E., Hiernaux, P., Kraus, D., Roupsard, O., Scheer, C., Srivastava, A. K., Tagesson, T., and Grote, R.: Modelling Gas Exchange and Biomass Production in West African Sahelian and Sudanian Ecological Zones. Karlsruhe Institute of Technology (KIT) [data set], https://doi.org/10.35097/437, 2021.
Roupsard, O., Audebert, A., Ndour, A. P., Clermont-Dauphin, C., Agbohessou, Y., Sanou, J., Koala, J., Faye, E., Sambakhe, D., Jourdan, C., le Maire, G., Tall, L., Sanogo, D., Seghieri, J., Cournac, L., and Leroux, L.: How far does the tree affect the crop in agroforestry? New spatial analysis methods in a Faidherbia parkland, Agr. Ecosyst. Environ., 296, 106928, https://doi.org/10.1016/j.agee.2020.106928, 2020.
Running, S. W. and Coughlan, J. C.: A general model of forest ecosystem processes for regional applications. I. Hydrologic balance, canopy gas exchange and primary production processes, Ecol. Model., 42, 125–154, https://doi.org/10.1016/0304-3800(88)90112-3, 1988.
Saleska, S. R., Miller, S. D., Matross, D. M., Goulden, M. L., Wofsy, S. C., da Rocha, H. R., de Camargo, P. B., Crill, P., Daube, B. C., de Freitas, H. C., Hutyra, L., Keller, M., Kirchhoff, V., Menton, M., Munger, J. W., Pyle, E. H., Rice, A. H., and Silva, H.: Carbon in amazon forests: Unexpected seasonal fluxes and disturbance-induced losses, Science, 302, 1554–1557, https://doi.org/10.1126/science.1091165, 2003.
Scheiter, S. and Higgins, S. I.: Impacts of climate change on the vegetation of Africa: an adaptive dynamic vegetation modelling approach, Glob. Change Biol., 15, 2224–2246, https://doi.org/10.1111/j.1365-2486.2008.01838.x, 2009.
Scholes, R. J. and Hall, D. O.: The carbon budget of tropical savannas, woodlands and grasslands, in: Global change: effects on coniferous forests and grasslands, edited by: Breymeyer, A. I., Hall, D. O., Melillo, J. M., and Ågren, G. I., Scope, John Wiley and Sons, Chichester, 69–100, 1996.
Sellers, P. J., Randall, D. A., Collatz, G. J., Berry, J. A., Field, C. B., Dazlich, D. A., Zhang, C., Collelo, G. D., and Bounoua, L.: A revised land surface parameterization (SiB2) for atmospheric GCMs. Part I: model formulation, J. Climate, 9, 676–705, https://doi.org/10.1175/1520-0442(1996)009<0676:ARLSPF>2.0.CO;2, 1996.
Setterfield, S. A., Clifton, P. J., Hutley, L. B., Rossiter-Rachor, N. A., and Douglas, M. M.: Exotic grass invasion alters microsite conditions limiting woody recruitment potential in an Australian savanna, Sci. Rep.-UK, 8, 6628–6628, https://doi.org/10.1038/s41598-018-24704-5, 2018.
Sibret, T.: The Sahelian Drylands under Pressure: Studying the Impact of Environmental Factors on Vegetation in Dahra, Senegal, Master of Science in Bioscience Engineering, Ghent University, Ghent, 113 pp., 2018.
Simioni, G., Le Roux, X., Gignoux, J., and Sinoquet, H.: Treegrass: a 3D, process-based model for simulating plant interactions in tree-grass ecosystems, Ecol. Model., 131, 47–63, https://doi.org/10.1016/S0304-3800(00)00243-X, 2000.
Sjöström, M., Ardö, J., Arneth, A., Boulain, N., Cappelaere, B., Eklundh, L., de Grandcourt, A., Kutsch, W. L., Merbold, L., Nouvellon, Y., Scholes, R. J., Schubert, P., Seaquist, J., and Veenendaal, E. M.: Exploring the potential of MODIS EVI for modeling gross primary production across African ecosystems, Remote. Sens. Environ., 115, 1081–1089, https://doi.org/10.1016/j.rse.2010.12.013, 2011.
Sjöström, M., Zhao, M., Archibald, S., Arneth, A., Cappelaere, B., Falk, U., de Grandcourt, A., Hanan, N., Kergoat, L., Kutsch, W., Merbold, L., Mougin, E., Nickless, A., Nouvellon, Y., Scholes, R. J., Veenendaal, E. M., and Ardö, J.: Evaluation of MODIS gross primary productivity for Africa using eddy covariance data, Remote. Sens. Environ., 131, 275–286, https://doi.org/10.1016/j.rse.2012.12.023, 2013.
Snyman, H. A.: Rangeland degradation in a semi-arid South Africa – I: influence on seasonal root distribution, root/shoot ratios and water-use efficiency, J. Arid Environ., 60, 457–481, https://doi.org/10.1016/j.jaridenv.2004.06.006, 2005.
Sobamowo, J. O.: Effect of harvesting dates and fertilizer application on cassava productivity in rainforest and guinea savanna agroecological zones of Nigeria, PhD, University of Cape Coast, Ghana, 2016.
Sonawane, B. V., Sharwood, R. E., von Caemmerer, S., Whitney, S. M., and Ghannoum, O.: Short-term thermal photosynthetic responses of C4 grasses are independent of the biochemical subtype, J. Exp. Bot., 68, 5583–5597, https://doi.org/10.1093/jxb/erx350, 2017.
Sotelo Montes, C., Weber, J. C., Silva, D. A., Andrade, C., Muñiz, G. B., Garcia, R. A., and Kalinganire, A.: Growth and fuelwood properties of five tree and shrub species in the Sahelian and Sudanian ecozones of Mali: relationships with mean annual rainfall and geographical coordinates, New Forests, 45, 179–197, https://doi.org/10.1007/s11056-013-9401-9, 2014.
Tagesson, T., Fensholt, R., Guiro, I., Rasmussen, M. O., Huber, S., Mbow, C., Garcia, M., Horion, S., Sandholt, I., Holm-Rasmussen, B., Göttsche, F. M., Ridler, M.-E., Olén, N., Lundegard Olsen, J., Ehammer, A., Madsen, M., Olesen, F. S., and Ardö, J.: Ecosystem properties of semiarid savanna grassland in West Africa and its relationship with environmental variability, Glob. Change Biol., 21, 250–264, https://doi.org/10.1111/gcb.12734, 2015.
Tagesson, T., Ardö, J., Guiro, I., Cropley, F., Mbow, C., Horion, S., Ehammer, A., Mougin, E., Delon, C., Corinne, G.-L., and Fensholt, R.: Very high CO2 exchange fluxes at the peak of the rainy season in a West African grazed semi-arid savanna ecosystem, Geografisk Tidsskrift-Danish J. Geography, 116, 1–17, https://doi.org/10.1080/00167223.2016.1178072, 2016.
Tews, J. and Jeltsch, F.: Modelling the impact of climate change on woody plant population dynamics in South African savanna, BMC Ecology, 4, 1–12, https://doi.org/10.1186/1472-6785-4-17, 2004.
Tews, J., Esther, A., Milton, S. J., and Jeltsch, F.: Linking a population model with an ecosystem model: Assessing the impact of land use and climate change on savanna shrub cover dynamics, Ecol. Model., 195, 219–228, https://doi.org/10.1016/j.ecolmodel.2005.11.025, 2006.
Thornley, J. H. M.: Instantaneous canopy photosynthesis: Analytical expressions for sun and shade leaves based on exponential light decay down the canopy and an acclimated non-rectangular hyperbola for leaf photosynthesis, Ann. Bot., 89, 451–458, https://doi.org/10.1093/aob/mcf071, 2002.
Thornley, J. H. M. and Cannell, M. G. R.: Modelling the components of plant respiration: Representation and realism, Ann. Bot., 85, 55–67, https://doi.org/10.1006/anbo.1999.0997, 2000.
Thornthwaite, C. W.: An approach toward a rational classification of climate, Geogr. Rev., 38, 55, https://doi.org/10.2307/210739, 1948.
Timouk, F., Kergoat, L., Mougin, E., Lloyd, C. R., Ceschia, E., Cohard, J. M., Rosnay, P. d., Hiernaux, P., Demarez, V., and Taylor, C. M.: Response of surface energy balance to water regime and vegetation development in a Sahelian landscape, J. Hydrol., 375, 178–189, https://doi.org/10.1016/j.jhydrol.2009.04.022, 2009.
Ullmann, I.: Stomatal conductance and transpiration of Acacia under field conditions: similarities and differences between leaves and phyllodes, Trees-Struct. Funct., 3, 45–56, https://doi.org/10.1007/BF00202400, 1989.
Ünlü, M. and Steduto, P.: Comparison of Photosynthetic Water use Efficiency of Sweet Sorghum at Canopy and Leaf Scales, Turkish Journal of Agriculture and Forestry, 24, 519–526, 2000.
Velluet, C., Demarty, J., Cappelaere, B., Braud, I., Issoufou, H. B.-A., Boulain, N., Ramier, D., Mainassara, I., Charvet, G., Boucher, M., Chazarin, J.-P., Oï, M., Yahou, H., Maidaji, B., Arpin-Pont, F., Benarrosh, N., Mahamane, A., Nazoumou, Y., Favreau, G., and Seghieri, J.: Building a field- and model-based climatology of local water and energy cycles in the cultivated Sahel – annual budgets and seasonality, Hydrol. Earth Syst. Sci., 18, 5001–5024, https://doi.org/10.5194/hess-18-5001-2014, 2014.
Vico, G. and Porporato, A.: Modelling C3 and C4 photosynthesis under water-stressed conditions, Plant Soil, 313, 187–203, https://doi.org/10.1007/s11104-008-9691-4, 2008.
Vitasse, Y., Francois, C., Delpierre, N., Dufrene, E., Kremer, A., Chuine, I., and Delzon, S.: Assessing the effects of climate change on the phenology of European temperate trees, Agric. Forest Meteorol., 151, 969–980, 2011.
Vitkauskaitė, G. and Venskaitytė, L.: Differences between C3 (Hordeum vulgare L.) and C4 (Panicum miliaceum L.) plants with respect to their resistance to water deficit, Žemdirbystė (Agriculture), 98, 349–356, 2011.
Von Caemmerer, S.: Biochemical models of leaf photosynthesis, Technicques in Plant Sciences, CSIRO, Collingwood VIC 3066, Australia, 2000.
Vu, J. C. V.: Acclimation of peanut (Arachis hypogaea L.) leaf photosynthesis to elevated growth CO2 and temperature, Environ. Exp. Bot., 53, 85–95, https://doi.org/10.1016/j.envexpbot.2004.03.006, 2005.
Werner, C., Haas, E., Grote, R., Gauder, M., Graeff-Hönninger, S., Claupein, W., and Butterbach-Bahl, K.: Biomass production potential from Populus short rotation systems in Romania, GCB Bioenergy, 4, 642–653, https://doi.org/10.1111/j.1757-1707.2012.01180.x, 2012.
Wesolowski, T. and Rowinski, P.: Timing of bud burst and tree-leaf development in a multispecies temperate forest, Forest Ecol. Manage., 237, 387–393, 2006.
Whitley, R., Beringer, J., Hutley, L. B., Abramowitz, G., De Kauwe, M. G., Duursma, R., Evans, B., Haverd, V., Li, L., Ryu, Y., Smith, B., Wang, Y.-P., Williams, M., and Yu, Q.: A model inter-comparison study to examine limiting factors in modelling Australian tropical savannas, Biogeosciences, 13, 3245–3265, https://doi.org/10.5194/bg-13-3245-2016, 2016.
Whitley, R., Beringer, J., Hutley, L. B., Abramowitz, G., De Kauwe, M. G., Evans, B., Haverd, V., Li, L., Moore, C., Ryu, Y., Scheiter, S., Schymanski, S. J., Smith, B., Wang, Y.-P., Williams, M., and Yu, Q.: Challenges and opportunities in land surface modelling of savanna ecosystems, Biogeosciences, 14, 4711–4732, https://doi.org/10.5194/bg-14-4711-2017, 2017.
Yao, N. g. R. and Goué, B.: Water use efficiency of a cassava crop as affected by soil water balance, Agric. Forest Meteorol., 61, 187–203, https://doi.org/10.1016/0168-1923(92)90049-A, 1992.
Yu, G.-R., Zhuang, J., and Yu, Z.-L.: An attempt to establish a synthetic model of photosynthesis-transpiration based on stomatal behavior for maize and soybean plants grown in field, J. Plant Physiol., 158, 861–874, https://doi.org/10.1078/0176-1617-00177, 2001.
Yuan, W., Zhou, G., Wang, Y., Han, X., and Wang, Y.: Simulating phenological characteristics of two dominant grass species in a semi-arid steppe ecosystem, Ecol. Res., 22, 784–791, https://doi.org/10.1007/s11284-006-0318-z, 2007.